Extraction of flavonoids from Mucuna pruriens seeds by ultrasound: Evaluation of their antioxidant properties and toxicity in Caenorhabditis elegans
DOI:
https://doi.org/10.17268/sci.agropecu.2026.036Palavras-chave:
Velvet bean, ultrasound extraction, Box–Behnken design, antioxidant compounds, Caenorhabditis elegansResumo
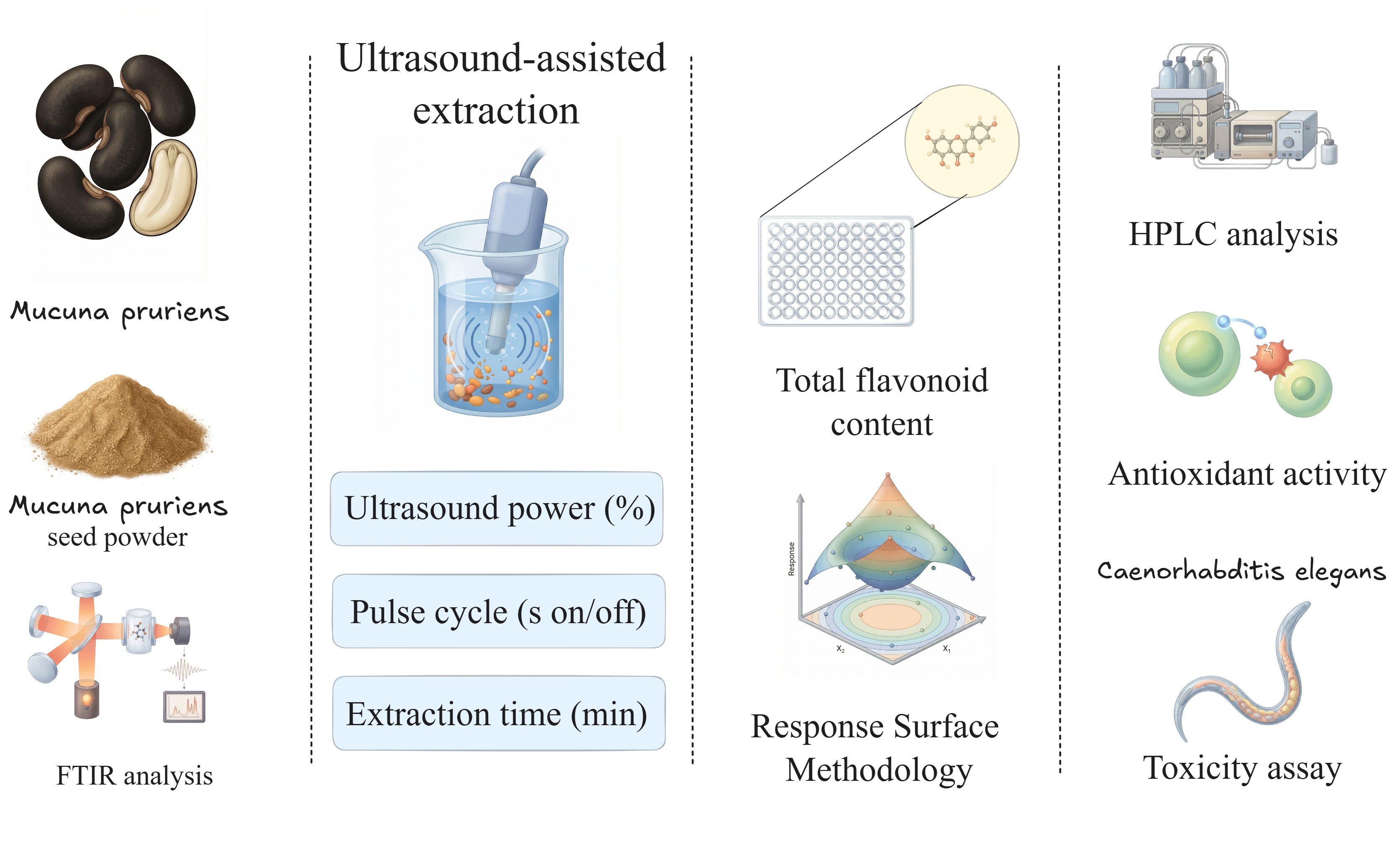
Traditional methods for extracting bioactive compounds from Mucuna pruriens involve high solvent use, long extraction times, and low yields. As a result, finding more efficient extraction techniques remains an important research focus. In this context, ultrasound-assisted extraction has become a promising alternative for extracting flavonoids from M. pruriens. These compounds play a crucial role in various applications, including nutraceuticals, pharmaceuticals, medicine, and cosmetics. Therefore, this study aimed to optimize the extraction yield of flavonoid content from Mucuna pruriens seeds by employing a Box-Behnken Design, focusing on the effects of ultrasound power, pulse cycle, and extraction time as factors, and using an acidified ethanol-water solution (80:20 v/v plus 2% HCl 2N) as the extraction solvent. Under optimal conditions, the ultrasound-assisted extraction method yielded a higher flavonoid content (77.30 µg/mL) than the magnetic stirring technique (60.92 µg/mL). High-performance liquid chromatography (HPLC) analysis identified and quantified significant amounts of gallocatechin, epicatechin, and epigallocatechin. Fourier-transform infrared (FTIR) spectroscopy suggested the presence of additional compounds, including amino acids, proteins, glycerides, aliphatic compounds, and alkaloids. Antioxidant assessments indicated that the optimal ultrasound conditions enhanced antioxidant activity in the DPPH and FRAP assays (323.51 and 259.07 mmol TE/g, respectively) compared to the conventional extraction method (294.13 and 164.19 mmol TE/g, respectively). Toxicity evaluations were conducted by exposing C. elegans to varying concentrations of extracts from both methods for 24 h, with LC50 values calculated at 143.65 µg/mL for the ultrasound extract under optimal conditions and 132.18 µg/mL for the magnetic stirring method. These findings provide support for the functional applications of Mucuna pruriens flavonoids as natural antioxidants.
Referências
Adefegha, S. A., Oboh, G., Oyeleye, S. I., Dada, F. A., Ejakpovi, I., & Boligon, A. A. (2017). Cognitive enhancing and antioxidative potentials of velvet beans (Mucuna pruriens) and horseradish (Moringa oleifera) seeds extracts: A comparative study. Journal of Food Biochemistry, 41(1), e12292. https://doi.org/10.1111/jfbc.12292
Aguilar-Hernández, G., García-Magaña, M. d. L., Vivar-Vera, M. d. l. Á., Sáyago-Ayerdi, S. G., Sánchez-Burgos, J. A., Morales-Castro, J., Anaya-Esparza, L. M., & Montalvo González, E. (2019). Optimization of ultrasound-assisted extraction of phenolic compounds from Annona muricata by-products and pulp. Molecules, 24(5), 904.
Anjaneyulu, J., Vidyashankar, R., & Godbole, A. (2020). Differential effect of Ayurvedic nootropics on C. elegans models of Parkinson’s disease. Journal of Ayurveda and integrative medicine, 11(4), 440-447. https://doi.org/10.1016/j.jaim.2020.07.006
Albarri, R., & Şahin, S. (2024). Monitoring the recovery of bioactive molecules from Moringa oleifera leaves: microwave treatment vs ultrasound treatment. Biomass Conversion and Biorefinery, 14(1), 1059-1071.
Anaya-Esparza, L. M., Aurora-Vigo, E. F., Villagrán, Z., Rodríguez-Lafitte, E., Ruvalcaba-Gómez, J. M., Solano-Cornejo, M. Á., Zamora-Gasga, V. M., Montalvo-González, E., Gómez-Rodríguez, H., Aceves-Aldrete, C. E., & González-Silva, N. (2023). Design of experiments for optimizing ultrasound-assisted extraction of bioactive compounds from plant-based sources. Molecules, 28(23), 7752. https://doi.org/10.3390/molecules28237752
Anaya-Esparza, L. M., Ramos-Aguirre, D., Zamora-Gasga, V. M., Yahia, E., & Montalvo-González, E. (2018). Optimization of ultrasonic-assisted extraction of phenolic compounds from Justicia spicigera leaves. Food Sci Biotechnol, 27(4), 1093-1102. https://doi.org/10.1007/s10068-018-0350-0
Avalos, B. I., Ojeda, G. A., Spinnenhirn, E. D., Acevedo, B. A., & Vallejos, M. M. (2023). Evaluation of phenolic compounds and antioxidant capacity in Mucuna pruriens and Cajanus cajan pods extracts. Food Chemistry Advances, 3, 100503. https://doi.org/10.1016/j.focha.2023.100503
Aware, C., Patil, R., Vyavahare, G., Gurav, R., Bapat, V., & Jadhav, J. (2019). Processing Effect on L-DOPA, In vitro protein and starch digestibility, proximate composition, and biological activities of promising legume: Mucuna macrocarpa. J Am Coll Nutr, 38(5), 447-456. https://doi.org/10.1080/07315724.2018.1547230
Bae, J., Kim, N., Shin, Y., Kim, S.-Y., & Kim, Y.-J. (2020). Activity of catechins and their applications. Biomedical Dermatology, 4(1), 8. https://doi.org/10.1186/s41702-020-0057-8
Benzie, I. F. F., & Strain, J. J. (1996). The rerric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Analytical Biochemistry, 239(1), 70-76. https://doi.org/10.1006/abio.1996.0292
Bhardwaj, P., Aiman, A., Iram, F., Saifi, I., Basir, S. F., Hassan, I., Islam, A., & Manzoor, N. (2024). Natural compound toxicity: An egregiously overlooked topic. In N. Manzoor (Ed.), Advances in Antifungal Drug Development: Natural Products with Antifungal Potential (pp. 653-673). Springer Nature Singapore. https://doi.org/10.1007/978-981-97-5165-5_25
Bitwell, C., Indra, S. S., Luke, C., & Kakoma, M. K. (2023). A review of modern and conventional extraction techniques and their applications for extracting phytochemicals from plants. Scientific African, 19, e01585. https://doi.org/10.1016/j.sciaf.2023.e01585
Chandrasiri Waliwita, W. A. L. (2021). Chapter 27 - Sri Lankan medicinal herbs used for the management of neurodegenerative diseases of the brain. In D. Ghosh (Ed.), Nutraceuticals in Brain Health and Beyond (pp. 379-389). Academic Press. https://doi.org/10.1016/B978-0-12-820593-8.00027-6
Chaudhary, S. K., De, A., Bhadra, S., & Mukherjee, P. K. (2015). Angiotensin-converting enzyme (ACE) inhibitory potential of standardized Mucuna pruriens seed extract. Pharm Biol, 53(11), 1614-1620. https://doi.org/10.3109/13880209.2014.996820
Chemat, F., Abert-Vian, M., Fabiano-Tixier, A. S., Strube, J., Uhlenbrock, L., Gunjevic, V., & Cravotto, G. (2019). Green extraction of natural products. Origins, current status, and future challenges. TrAC Trends in Analytical Chemistry, 118, 248-263. https://doi.org/10.1016/j.trac.2019.05.037
Chookiat, S., Theansungnoen, T., Kiattisin, K., & Intharuksa, A. (2024). Nanoemulsions containing Mucuna pruriens (L.) DC. seed extract for cosmetic applications. Cosmetics, 11(1), 29.
CONABIO. (2009). Stizolobium pruriens - ficha informativa. https://www.gob.mx/cms/uploads/attachment/file/221056/Mucuna_pruriens_utilis_final.pdf
Costa, A. C. C., Oliveira, L., Dorea, M. A., Leite, G., Amorim, R., Neto, E. D. S. M., Júnior, L. N., Silva, R. C., Silveira, E. L., & Fontelles, M. J. (2018). PO-029 Supercritical carbon dioxide extract of Mucuna pruriens inhibits brain cancer tumour growth in rats. ESMO Open, 3, A239. https://doi.org/10.1136/esmoopen-2018-EACR25.564
Dhanani, T., Singh, R., Shah, S., Kumari, P., & Kumar, S. (2015). Comparison of green extraction methods with conventional extraction method for extract yield, L-DOPA concentration and antioxidant activity of Mucuna pruriens seed. Green Chemistry Letters and Reviews, 8(2), 43-48. https://doi.org/10.1080/17518253.2015.1075070
Fernández-Barbero, G., Pinedo, C., Espada-Bellido, E., Ferreiro-González, M., Carrera, C., Palma, M., & García-Barroso, C. (2019). Optimization of ultrasound-assisted extraction of bioactive compounds from jabuticaba (Myrciaria cauliflora) fruit through a Box-Behnken experimental design. Food Science and Technology, 39(4), 1018-1029. https://doi.org/10.1590/fst.16918
Gallegos-Saucedo, M. d. J., Camargo-Hernández, G., Castillo-Romero, A., Ramírez-Herrera, M. A., Bañuelos-Pineda, J., Pereira-Suárez, A. L., Hernández-Chávez, A., & Hernández-Hernández, L. (2020). GABAergic system’s injuries induced by sodium sulfite in Caenorhabditis elegans were prevented by the anti-oxidative properties of dehydroepiandrosterone sulfate. Neurotoxicity Research, 38(2), 447-460. https://doi.org/10.1007/s12640-020-00207-y
Gao, S., Chen, W., Zeng, Y., Jing, H., Zhang, N., Flavel, M., Jois, M., Han, J.-D. J., Xian, B., & Li, G. (2018). Classification and prediction of toxicity of chemicals using an automated phenotypic profiling of Caenorhabditis elegans. BMC Pharmacology and Toxicology, 19(1), 18. https://doi.org/10.1186/s40360-018-0208-3
Hajibeglou, A., Sudagar, M., Azizinezhad-Jolandan, F., & Alaei, K. (2023). The effects of Mucuna pruriens and Basella alba extracts on sex reversal in rainbow trout (Oncorhynchus mykiss). Aquaculture Research, 2023, 1-6. https://doi.org/10.1155/2023/8252139
Han, D.-G., Bae, M.-J., & An, B.-J. (2022). Anti-inflammatory activity of velvet bean (Mucuna pruriens) substances in LPS−stimulated RAW 264.7 macrophages. Molecules, 27(24), 8797.
Hernández-Estrada, S., Anaya-Esparza, L. M., González-Torres, S., Hernández-Villaseñor, L. A., Gómez-Rodríguez, V. M., Ramírez-Vega, H., Villagrán, Z., Ruvalcaba-Gómez, J. M., Rodríguez-Barajas, N., & Montalvo-González, E. (2024). Extraction of soluble phenols and flavonoids from native Mexican pigmented corn kernel powder by ultrasound: Optimization process using response surface methodology. Applied Sciences, 14(17), 7869. https://doi.org/10.3390/app14177869
Hosseini, S. S., Khodaiyan, F., Kazemi, M., & Najari, Z. (2019). Optimization and characterization of pectin extracted from sour orange peel by ultrasound assisted method. International Journal of Biological Macromolecules, 125, 621-629. https://doi.org/10.1016/j.ijbiomac.2018.12.096
Iftikhar, M., Zhang, H., Iftikhar, A., Raza, A., Begum, N., Tahamina, A., Syed, H., Khan, M., & Wang, J. (2020). Study on optimization of ultrasonic assisted extraction of phenolic compounds from rye bran. LWT, 134, 110243. https://doi.org/10.1016/j.lwt.2020.110243
Intharuksa, A., Denduangboripant, J., Chansakaow, S., Thongkhao, K., & Sukrong, S. (2023). HPLC and DNA barcoding profiles for identification of the selected twelve Mucuna species and its application for detecting prohibited aphrodisiac Mucuna products. Heliyon, 9(3), e14130. https://doi.org/10.1016/j.heliyon.2023.e14130
Jabbar, S., Abid, M., Wu, T., Hashim, M. M., Saeeduddin, M., Hu, B., Lei, S., & Zeng, X. (2015). Ultrasound-assisted extraction of bioactive compounds and antioxidants from carrot pomace: A response surface approach. Journal of Food Processing and Preservation, 39(6), 1878-1888. https://doi.org/10.1111/jfpp.12425
Jiang, X., Liu, N., Liu, Y., Gao, L., & Xia, T. (2025). Exploration of the biosynthesis of galloylated catechins in tea plants. Beverage Plant Research, 5(1).
Jitpimai, K., Ngiwsara, L., Lang, W., Panichpat, T., Mingma, R., Svasti, J., & Wongchawalit, J. (2023). Evaluation of alcoholic extracts of Mucuna pruriens (L.) DC. var. utilis for antibacterial, antioxidant and cytotoxic activities toward human cancer cell lines. Chiang Mai Journal of Science, 50(5), 1-15.
Jimoh, M. A., Idris, O. A., & Jimoh, M. O. (2020). Cytotoxicity, phytochemical, antiparasitic screening, and antioxidant activities of Mucuna pruriens (Fabaceae). Plants (Basel), 9(9). https://doi.org/10.3390/plants9091249
Katiki, L. M., Ferreira, J. F. S., Gonzalez, J. M., Zajac, A. M., Lindsay, D. S., Chagas, A. C. S., & Amarante, A. F. T. (2013). Anthelmintic effect of plant extracts containing condensed and hydrolyzable tannins on Caenorhabditis elegans, and their antioxidant capacity. Veterinary Parasitology, 192(1), 218-227. https://doi.org/10.1016/j.vetpar.2012.09.030
Kedam, T. (2017). The traditional uses and pharmacological activities of Mucuna pruriens (L)DC: A comprehensive review. Indo American Journal of Pharmaceutical Research, 7, 7516.
Khorasani Esmaeili, A., Mat Taha, R., Mohajer, S., & Banisalam, B. (2015). Antioxidant activity and total phenolic and flavonoid content of various solvent extracts from in vivo and in vitro grown Trifolium pratense L. (Red Clover). Biomed Res Int, 2015, 643285. https://doi.org/10.1155/2015/643285
Kobus, Z., Krzywicka, M., Starek-Wójcicka, A., & Sagan, A. (2022). Effect of the duty cycle of the ultrasonic processor on the efficiency of extraction of phenolic compounds from Sorbus intermedia. Scientific reports, 12(1), 8311. https://doi.org/10.1038/s41598-022-12244-y
Kulkarni, S. R., Thokchom, B., Abbigeri, M. B., Bhavi, S. M., Singh, S. R., Metri, N., & Yarajarla, R. B. (2025). The role of L-DOPA in neurological and neurodegenerative complications: a review. Molecular and Cellular Biochemistry, 1-22.
Kumar, A., & Srinivasa Rao, P. (2020). Optimization of pulsed-mode ultrasound assisted extraction of bioactive compounds from pomegranate peel using response surface methodology. Journal of Food Measurement and Characterization, 14(6), 3493-3507. https://doi.org/10.1007/s11694-020-00597-9
Leite, N. R., de Araújo, L. C. A., Dos Santos da Rocha, P., Agarrayua, D. A., Ávila, D. S., Carollo, C. A., Silva, D. B., Estevinho, L. M., de Picoli Souza, K., & Dos Santos, E. L. (2020). Baru Pulp (Dipteryx alata Vogel): Fruit from the brazilian savanna protects against oxidative stress and increases the life expectancy of Caenorhabditis elegans via SOD-3 and DAF-16. Biomolecules, 10(8). https://doi.org/10.3390/biom10081106
Maillot, A., Schmitt, C., & Marteau, A. (2022). Poisoning after ingestion of Mucuna pruriens Seeds on Reunion Island. Wilderness Environ Med, 33(1), 122-124. doi:10.1016/j.wem.2021.10.004
Mita, S. R., Husni, P., Putriana, N. A., Maharani, R., Hendrawan, R. P., & Dewi, D. A. (2024). A recent update on the potential use of catechins in cosmeceuticals. Cosmetics, 11(1), 23.
Molina-Hernández, J. B., Scroccarello, A., Della Pelle, F., De Flaviis, R., Compagnone, D., Del Carlo, M., Paparella, A., & Chaves Lόpez, C. (2022). Synergistic antifungal activity of catechin and silver nanoparticles on Aspergillus niger isolated from coffee seeds. LWT, 169, 113990. https://doi.org/10.1016/j.lwt.2022.113990
Ndayiragije, E., Caumul, P., Joondan, N., Bhowon, M. G., & Laulloo, S. J. (2024). l-Tyrosine and l-DOPA: Promising scaffolds for the synthesis of biologically active compounds. Comptes Rendus. Chimie, 27(G1), 299-317.
Nwaoguikpe, R., Braide, W., & Ujowundu, C. (2011). The effects of processing on the proximate and phytochemical compositions of Mucuna pruriens seeds (velvet beans). Pakistan Journal of Nutrition, 10(10), 947-951.
Parvatikar, P. P., Patil, S. M., Patil, B. S., Reddy, R. C., Bagoji, I., Kotennavar, M. S., Patil, S., Patil, A. V., Das, K. K., Das, S. N., & Bagali, S. (2023). Effect of Mucuna pruriens on brain NMDA receptor and tau protein gene expression in cerebral ischemic rats. Frontiers in Physiology, 14. https://doi.org/10.3389/fphys.2023.1092032
Pathania, R., Chawla, P., Khan, H., Kaushik, R., & Khan, M. A. (2020). An assessment of potential nutritive and medicinal properties of Mucuna pruriens: a natural food legume. 3 Biotech, 10(6). doi:10.1007/s13205-020-02253-x
Pérez-Jiménez, J., Arranz, S., Tabernero, M., Díaz- Rubio, M. E., Serrano, J., Goñi, I., & Saura-Calixto, F. (2008). Updated methodology to determine antioxidant capacity in plant foods, oils and beverages: Extraction, measurement and expression of results. Food Research International, 41(3), 274-285. https://doi.org/10.1016/j.foodres.2007.12.004
Prior, R. L., Wu, X., & Schaich, K. (2005). Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. Journal of Agricultural and Food Chemistry, 53(10), 4290-4302. https://doi.org/10.1021/jf0502698
Rachsee, A., Chiranthanut, N., Kunnaja, P., Sireeratawong, S., Khonsung, P., Chansakaow, S., & Panthong, A. (2021). Mucuna pruriens (L.) DC. seed extract inhibits lipopolysaccharide-induced inflammatory responses in BV2 microglial cells. J Ethnopharmacol, 267, 113518. https://doi.org/10.1016/j.jep.2020.113518
Rheem, S. (2023). Optimizing food processing through a new approach to response surface methodology. Food science of animal resources, 43(2), 374-381. https://doi.org/10.5851/kosfa.2023.e7
Rangsinth, P., Prasansuklab, A., Duangjan, C., Gu, X., Meemon, K., Wink, M., & Tencomnao, T. (2019). Leaf extract of Caesalpinia mimosoides enhances oxidative stress resistance and prolongs lifespan in Caenorhabditis elegans. BMC Complementary and Alternative Medicine, 19(1). https://doi.org/10.1186/s12906-019-2578-5
Rashad, S., El-Chaghaby, G., Lima, E. C., & Simoes Dos Reis, G. (2023). Optimizing the ultrasonic-assisted extraction of antioxidants from Ulva lactuca algal biomass using factorial design. Biomass Conversion and Biorefinery, 13(7), 5681-5690. https://doi.org/10.1007/s13399-021-01516-8
Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., & Rice-Evans, C. (1999). Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine, 26(9), 1231-1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Rima, Ishmayana, S., Made Malini, D., & Soedjanaatmadja, U. M. S. (2023). Nutritional content and the activities of l-Dopa (L-3,4-dihydoxyphenyalanine) from Mucuna pruriens L. DC seeds of Central Java accession. Arabian Journal of Chemistry, 16(1), 104390. https://doi.org/10.1016/j.arabjc.2022.104390
Sang, X., Zhen, F., Lv, P., Zhang, Z., Qu, B., & Wang, Y. (2025). Green and chemical-free pretreatment of flavonoids in tea plant seed husk using ultrasound-cold isostatic pressure synergistic extraction. Food Chemistry, 478, 143725. https://doi.org/10.1016/j.foodchem.2025.143725
Sayem, A. S. M., Ahmed, T., Mithun, M. U. K., Rashid, M., & Rana, M. R. (2024). Optimising ultrasound-assisted extraction conditions for maximising phenolic, flavonoid content and antioxidant activity in hog plum peel and seed: A response surface methodology approach. Journal of Agriculture and Food Research, 18, 101312. https://doi.org/10.1016/j.jafr.2024.101312
Sowdhanya, D., Singh, J., Rasane, P., Kaur, S., Kaur, J., Ercisli, S., & Verma, H. (2024). Nutritional significance of velvet bean (Mucuna pruriens) and opportunities for its processing into value-added products. Journal of Agriculture and Food Research, 15, 100921. https://doi.org/10.1016/j.jafr.2023.100921
Taroncher, M., Vila-Donat, P., Tolosa, J., Ruiz, M. J., & Rodríguez-Carrasco, Y. (2021). Biological activity and toxicity of plant nutraceuticals: an overview. Current Opinion in Food Science, 42, 113-118. doi:https://doi.org/10.1016/j.cofs.2021.05.008
Tavares, R. l., Vasconcelos, M. H. A. d., Dutra, M. L. d. V., D’Oliveira, A. B., Lima, M. d. S., Salvadori, M. G. d. S. S., Pereira, R. d. A., Alves, A. F., Nascimento, Y. M. d., Tavares, J. F., Guzman-Quevedo, O., & Aquino, J. d. S. (2020). Mucuna pruriens administration minimizes neuroinflammation and shows anxiolytic, antidepressant and slimming effects in obese rats. Molecules, 25(23), 5559.
Tesoro, C., Lelario, F., Ciriello, R., Bianco, G., Di Capua, A., & Acquavia, M. A. (2022). An overview of methods for L-dopa extraction and analytical determination in plant matrices. Separations, 9(8), 224. https://doi.org/10.3390/separations9080224
Vilairat, C., Kobtrakul, K., & Vimolmangkang, S. (2023). Enhanced physicochemical stability of the L-DOPA extract of Mucuna pruriens seeds by adding Phyllanthus emblica. Molecules, 28(4), 1573.
Vinatoru, M., Mason, T. J., & Calinescu, I. (2017). Ultrasonically assisted extraction (UAE) and microwave assisted extraction (MAE) of functional compounds from plant materials. TrAC Trends in Analytical Chemistry, 97, 159-178. doi:https://doi.org/10.1016/j.trac.2017.09.002
Widaad, A., Zulkipli, I. N., & Petalcorin, M. I. R. (2022). Anthelmintic effect of Leucaena leucocephala extract and its active compound, mimosine, on vital behavioral activities in Caenorhabditis elegans. Molecules, 27(6), 1875. Retrieved from https://www.mdpi.com/1420-3049/27/6/1875
Xiang, Y., Liu, Z., Liu, Y., Dong, B., Yang, C., & Li, H. (2024). Ultrasound-assisted extraction, optimization, and purification of total flavonoids from Daphnegenkwa and analysis of their antioxidant, anti-inflammatory, and analgesic activities. Ultrasonics Sonochemistry, 111, 107079. https://doi.org/10.1016/j.ultsonch.2024.107079
Yadav, M., Upadhyay, P., Purohit, P., Pandey, B., & Shah, H. (2017). Phytochemistry and pharmacological activity of Mucuna pruriens: A review. International Journal of Green Pharmacy, 11, 69-73. https://doi.org/10.22377/ijgp.v11i02.916
Yadav, R. K., Yadav, R. A., & Kostova, I. (2023). Structural, conformational and spectroscopic investigations of a biologically active compound: L-Dopa. Applied Sciences, 13(24), 13336.
Zarroug, S. H. O., Bajaman, J. S., Hamza, F. N., Saleem, R. A., & Abdalla, H. K. (2023). Caenorhabditis elegans as an in vivo model for the discovery and development of natural plant-based antimicrobial compounds. Pharmaceuticals, 16(8), 1070. https://doi.org/10.3390/ph16081070
Downloads
Arquivos adicionais
Publicado
Como Citar
Edição
Seção
Licença
Copyright (c) 2026 Scientia Agropecuaria

Este trabalho está licenciado sob uma licença Creative Commons Attribution-NonCommercial 4.0 International License.
Los autores que publican en esta revista aceptan los siguientes términos:
a. Los autores conservan los derechos de autor y conceden a la revista el derecho publicación, simultáneamente licenciada bajo una licencia de Creative Commons que permite a otros compartir el trabajo, pero citando la publicación inicial en esta revista.
b. Los autores pueden celebrar acuerdos contractuales adicionales separados para la distribución no exclusiva de la versión publicada de la obra de la revista (por ejemplo, publicarla en un repositorio institucional o publicarla en un libro), pero citando la publicación inicial en esta revista.
c. Se permite y anima a los autores a publicar su trabajo en línea (por ejemplo, en repositorios institucionales o en su sitio web) antes y durante el proceso de presentación, ya que puede conducir a intercambios productivos, así como una mayor citación del trabajo publicado (ver efecto del acceso abierto).




