Endophytic bacteria isolated from the medicinal plant kratom (Mitragyna speciosa): Morphological, biochemical, and molecular characteristics, and their potential use as plant growth promoters
DOI:
https://doi.org/10.17268/sci.agropecu.2026.026Palavras-chave:
Endophytic bacteria, kratom, Mitragyna speciosa, plant growth-promoting, qualitative and quantitative assaysResumo
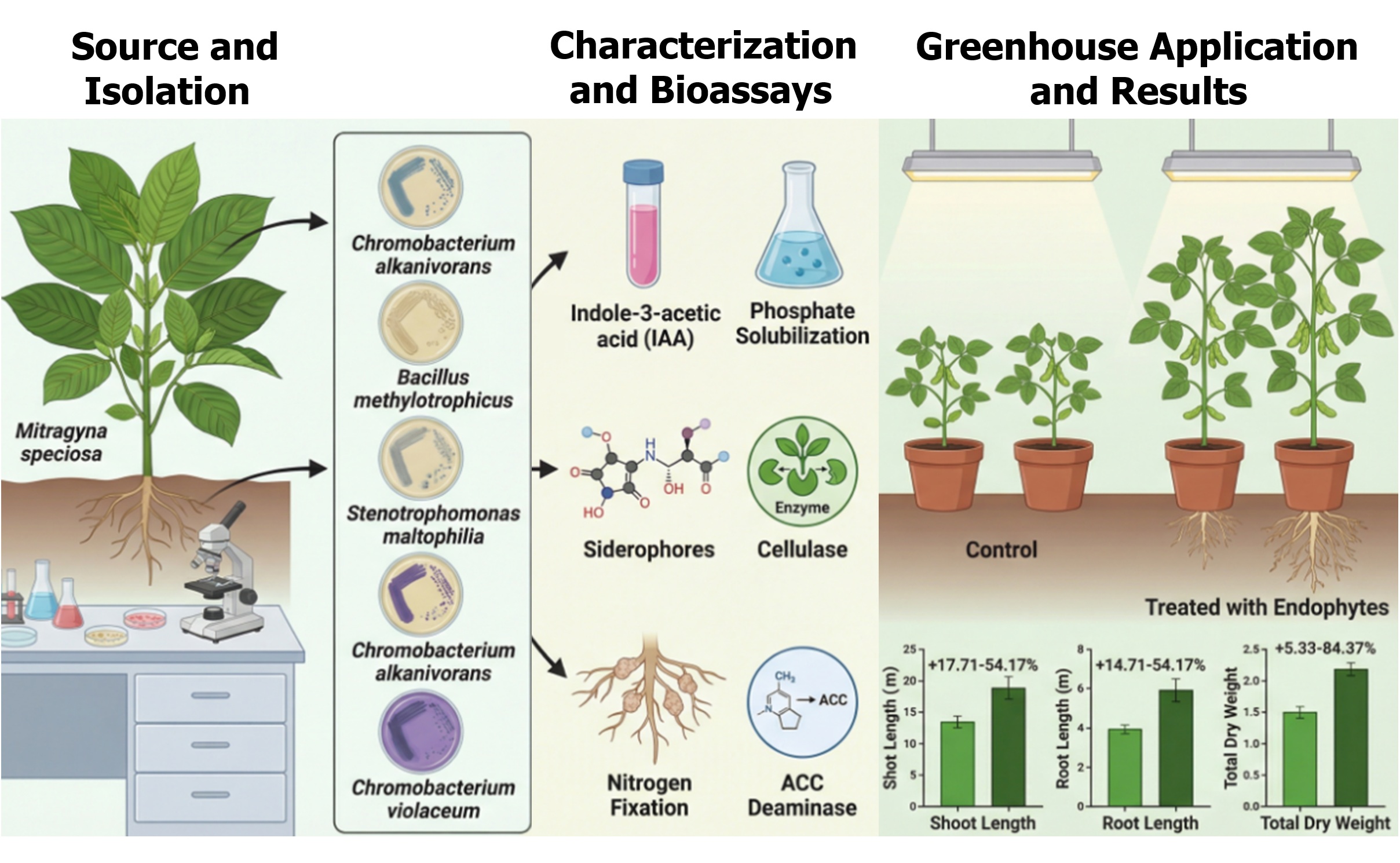
Endophytic bacteria live within and colonize plant tissues, forming symbiotic interactions that often enhance host growth, health, and stress tolerance without causing harm. This study aimed to isolate endophytic bacterial strains from the medicinal plant ‘kratom’ (Mitragyna speciosa), characterize their biological functions by analyzing morphological, biochemical, and molecular properties, and evaluate their plant growth-promoting potential. The research followed standard procedures for isolating and analyzing the production of plant growth-promoting compounds, including indole-3-acetic acid, 1-aminocyclopropane-1-carboxylate deaminase, siderophores, cellulase, nitrogen fixation, and phosphate solubilization, and for assessing their effects on soybean in planta, including bacterial root colonization and plant growth. Of the isolates tested by 16S rDNA sequencing, five were successfully identified, belonging to Chromobacterium alkanivorans (2), Bacillus methylotrophicus (1), Stenotrophomonas maltophilia (1), and Chromobacterium violaceum (1). Quantitative assays showed that the bacterial isolates produced 13.35 – 24.11 g mL-1 of indole acetic acid, 20.77 – 24.95 μg mL-1 of inorganic phosphate solubilization activity, 63.34 – 84.29 U mL-1 of acid phosphatase, and 2.92 – 4.86 U mL-1 of cellulase. Qualitative tests confirmed that each isolate produced siderophores, nitrogenase, 1-aminocyclopropane-1-carboxylate deaminase, indole acetic acid, cellulase, and both organic and inorganic phosphate solubilization. Greenhouse experiments demonstrated that all bacterial isolates promoted plant growth, increasing soybean shoot length by 17.71% to 54.17%, root length by 14.36% to 39.34%, and total dry weight by 2.24% to 56.71% compared to controls. These findings support the potential application of endophytes as biofertilizers in sustainable agriculture, highlighting their capacity as plant growth-promoting bacteria.
Referências
Achari, S. R., Kaur, J., Dinh, Q., & others. (2020). Phylogenetic relationship between Australian Fusarium oxysporum isolates and resolving the species complex using the multispecies coalescent model. BMC Genomics, 21, 248. https://doi.org/10.1186/s12864-020-6640-y
Acuña, R., Rouard, M., Leiva, A. M., Marques, C., Olortegui, J. A., Ureta, C., & Dita, M. (2022). First report of Fusarium oxysporum f. Sp. Cubense tropical race 4 causing fusarium wilt in Cavendish bananas in Peru. Plant Disease, 106(8), 2268. https://doi.org/10.1094/pdis-09-21-1951-pdn
Agrios, G. N. (2005). Plant pathology (5th ed.). Elsevier Academic Press.
Arie, T. (2019). Fusarium diseases of cultivated plants, control, diagnosis, and molecular and genetic studies. Journal of Pesticide Science, 44(4), 275–281. https://doi.org/10.1584/jpestics.J19-03
Asociación de Exportadores de Banano del Ecuador (AEBE). (2025). Sitio web de la AEBE. https://www.aebe.com.ec/
Ayuso-Álvarez, A., García-Pérez, J., Triviño-Juárez, J.-M., Larrinaga-Torrontegui, U., González-Sánchez, M., Ramis, R., Boldo, E., López-Abente, G., Galán, I., & Fernández-Navarro, P. (2020). Association between proximity to industrial chemical installations and cancer mortality in Spain. Environmental Pollution, 260, 113869. https://doi.org/https://doi.org/10.1016/j.envpol.2019.113869
Bakhat, N., Vielba-Fernández, A., Padilla-Roji, I., Martínez-Cruz, J., Polonio, Á., Fernández-Ortuño, D., & Pérez-García, A. (2023). Suppression of Chitin-Triggered Immunity by Plant Fungal Pathogens: A Case Study of the Cucurbit Powdery Mildew Fungus Podosphaera xanthii. Journal of Fungi, 9(7), 771. https://doi.org/10.3390/jof9070771
Balajee, S. A., Borman, A. M., Brandt, M. E., & others. (2009). Sequence-based identification of Aspergillus, Fusarium, and Mucorales species in the clinical mycology laboratory: where are we and where should we go from here. Journal of Clinical Microbiology. https://doi.org/10.1128/jcm.01685-08
Baruah, A., Bora, P., Damodaran, T., & others. (2025). Patho-Ecological Distribution and Genetic Diversity of Fusarium oxysporum f. sp. cubense in Malbhog Banana Belts of Assam, India. Journal of Fungi, 11, 195. https://doi.org/10.20944/preprints202409.1184.v1
Dita, M., Barquero, M., Heck, D., Mizubuti, E. S. G., & Staver, C. P. (2018). Fusarium Wilt of Banana: Current Knowledge on Epidemiology and Research Needs Toward Sustainable Disease Management. Frontiers in Plant Science, 9, 1468. https://doi.org/10.3389/fpls.2018.01468
Drenth, A., & Kema, G. (2021). The Vulnerability of Bananas to Globally Emerging Disease Threats. Phytopathology, 111(12), 2146–2161. https://doi.org/10.1094/PHYTO-07-20-0311-RVW
FAO. (2020). Perspectiva a mediano plazo de la producción y el comercio mundial de bananos y frutas tropicales.
FAO. (2025). Disponible online.
FAOSTAT. (2023). FAOSTAT Statistical Database.
García, L., Grajales, A., Cárdenas, M. E., Sierra, R., Lozano, G., Garavito, M., & others. (2011). Isolation and characterization of two strains of Fusarium oxysporum causing potato dry rot in Solanum tuberosum in Colombia. Rev Iberoam Micol, 28(4), 166–172.
García-Bastidas, F., Van Der Veen, A. J. T., Nakasato-Tagami, G., Meijer, H. J. G., Arango-Isaza, R. E., & Kema, G. H. J. (2019). An improved phenotyping protocol for Panama disease in banana. Frontiers in Plant Science. https://doi.org/10.3389/fpls.2019.01006
Leslie, J. F., & Summerell, B. A. (2006). The Fusarium Laboratory Manual. Blackwell Publishing. https://doi.org/10.1002/9780470278376
Ma, L. J., Geiser, D. M., Proctor, R. H., Rooney, A. P., O’Donnell, K., Trail, F., Gardiner, D. M., Manners, J. M., & Kazan, K. (2013). Fusarium pathogenomics. Annual Review of Microbiology, 67, 399–416. https://doi.org/10.1146/annurev-micro-092412-155650
Magdama, F., Magdama, F., Monserrate-Maggi, L., Serrano, L., Sosa, D., Geiser, D. M., & Jiménez-Gasco, M. del M. (2019). Comparative analysis uncovers the limitations of current molecular detection methods for Fusarium oxysporum f. sp. cubense race 4 strains. PLOS ONE, 14(9). https://doi.org/10.1371/journal.pone.0222727
Martínez, G., Olivares, B. O., Rey, J. C., Rojas, J., Cardenas, J., Muentes, C., & Dawson, C. (2023). The advance of Fusarium wilt tropical race 4 in musaceae of Latin America and the Caribbean: current situation. Pathogens, 12(2), 277. https://doi.org/10.3390/pathogens12020277
Mejías Herrera, R., Hernández, Y., Magdama, F., Mostert, D., Bothma, S., Paredes, E. M., Terán, D., González, E., Angulo, R., Angel, L., Rodríguez, Y., Ortega, R., Viljoen, A., & Marys, E. E. D. R. (2022). First report of Fusarium wilt of cavendish bananas caused by Fusarium oxysporum f. Sp. Cubense tropical race 4 in Venezuela. Plant Disease. https://doi.org/10.1094/pdis-04-23-0781-pdn
Nozawa, S., Seto, Y., Takata, Y., Narreto, L. A., Valle, R. R., Okui, K., Taida, S., Alvindia, D. G., Reyes, R. G., & Watanabe, K. (2023). Fusarium mindanaoense sp. nov., a New Fusarium Wilt Pathogen of Cavendish Banana from the Philippines Belonging to the F. fujikuroi Species Complex. Journal of Fungi, 9(4), 443. https://doi.org/10.3390/jof9040443
O’Donnell, K., Ward, T., Robert, V., Crous, P., Geiser, D., & Kang, S. (2015). DNA sequence-based identification of Fusarium: current status and future directions. Phytoparasitica, 43, 583–595. https://doi.org/10.1007/s12600-015-0484-z
Olivares, B. O., Rey, J. C., Lobo, D., Navas-Cortés, J. A., Gómez, J. A., & Landa, B. B. (2021). Marchitez por Fusarium del banano: una revisión de los factores agroambientales del sistema productivo venezolano que afectan su desarrollo. Agronomía, 11(5), 986. https://doi.org/10.3390/agronomy11050986
Pérez-Vicente, L., Dita, M. A., & la Parte, E. (2014). Prevención y diagnóstico de la marchitez por Fusarium (mal de Panamá) del banano causada por Fusarium oxysporum f. sp. cubense. Raza Tropical 4 (RT4).
Ploetz, R. C. (2000). Panama disease: A classic and destructive disease of banana. Plant Health Progress. https://doi.org/10.1094/PHP-2000-1204-01-HM
Ploetz, R. C., Kema, G. H. J., & Ma, L.-J. (2015). Impact of diseases on export and smallholder production of banana. Annual Review of Phytopathology, 53, 269–288.
Purwati, R., Hidayah, N., Sudjindro, & Sudarsono. (2008). Inoculation methods and conidial densities of Fusarium oxysporum f.sp. cubense in abaca. Hayati Journal of Biosciences, 15(1), 1–7. https://doi.org/10.4308/hjb.15.1.1
Rodríguez-Yzquierdo, G., Olivares, B. O., González-Ulloa, A., León-Pacheco, R., Gómez-Correa, J. C., Yacomelo-Hernández, M., Carrascal-Pérez, F., Florez-Cordero, E., Soto-Suárez, M., Dita, M., & Betancourt-Vásquez, M. (2023). Soil predisposing factors to Fusarium oxysporum f.sp cubense tropical race 4 on banana crops of la Guajira, Colombia. Agronomy, 13(10), 2588. https://doi.org/10.3390/agronomy13102588
Singha, I. M., Kakoty, Y., Unni, B. G., Das, J., & Kalita, M. C. (2016). Identification and characterization of Fusarium sp. Using its and RAPD causing Fusarium wilt of tomato isolated from Assam, North East India. Journal of Genetic Engineering and Biotechnology. https://doi.org/10.1016/j.jgeb.2016.07.001
Ferreira, D. F. (2019). SISVAR: A computer analysis system to fixed to effects split plot type designs. Brazilian Journal of Biometrics, 37(4), 529–535. https://doi.org/10.28951/rbb.v37i4.450
Strobl, E., & Mohan, P. (2020). Climate and the Global Spread and Impact of Bananas’ Black Leaf Sigatoka Disease. Atmosphere, 11(9), 947. https://doi.org/10.3390/atmos11090947
Su, Y., Liu, C., Fang, H., & Zhang, D. (2020). Bacillus subtilis: a universal cell factory for industry, agriculture, biomaterials and medicine. Microbial Cell Factories, 19, 173. https://doi.org/10.1186/s12934-020-01436-8
Validov, S. Z., Kamilova, F. D., & Lugtenberg, B. J. (2011). Monitoring of pathogenic and non-pathogenic Fusarium oxysporum Abdillah., Bardgett, R.D., & van der Putten, W.H. (2014). Belowground biodiversity and ecosystem functioning. Nature, 515(7528), 505–511. https://doi.org/10.1038/nature13855
Afzal, I., Shinwari, Z.K., Sikandar, S., & Shahzad, S. (2019). Plant beneficial endophytic bacteria: Mechanisms, diversity, host range and genetic determinants. Microbiological Research, 221, 36–49. https://doi.org/10.1016/j.micres.2019.02.001.
AL Kahtani, M.D.F., Fouda, A., Attia, K.A., Eid, A.M., Ewais, E.E., Hijri, M., Hassan, S. E., Khan, N., Hafez, Y.M., & Abdelaal, K.A. (2020). Isolation and characterization of plant growth promoting endophytic bacteria from desert plants and their application as bioinoculants for sustainable agriculture. Agronomy, 10(9), 1325.https://doi.org/10.3390/agronomy10091325
Angel, R., Nepel, M., Panhölzl, C., Schmidt, H., Herbold, C. W., Eichorst, S. A., & Woebken, D. (2018). Evaluation of primers targeting the diazotroph functional gene and development of nif map a bioinformatics pipeline for analyzing nifh amplicon data. Frontiers in Microbiology, 9, 703.https://doi.org/10.3389/fmicb.2018.00703
Anillo, H.J.B., Sanchez, C.J.O., Lima, G.D.S., & dos Santos, G.F. (2016). Endophytic microorganisms isolated of plants grown in Colombia: a short review. Journal of Microbial Biochemical Technology, 8(8), 509-513. https://doi.org/10.4172/1948-5948.1000335
Auld, R.R., Myre, M., Mykytczuk, N.C., Leduc, L.G., & Merritt, T.J. (2013). Characterization of the microbial acid mine drainage microbial community using culturing and direct sequencing techniques. Journal of Microbiological Methods, 93(2), 108–115.https://doi.org/10.1016/j.mimet.2013.01.023
Aydi Ben Abdallah, R., Jabnoun-Khiareddine, H., Nefzi, A., & Daami-Remadi, M. (2018). Evaluation of the growth-promoting potential of endophyticbacteria recovered from healthy tomato plants. Journal of Horticulture, 5, 234. https://www.tjpp.tn
Behera, B.C., Yadav, H., Singh, S.K., Sethi, B.K., Mishra, R.R., Kumari, S., & Thatoi, H. (2016). Alkaline phosphatase activity of a phosphate solubilizing Alcaligenes faecalis, isolated from Mangrove soil. Biotechnology Research and Innovation, 1(1), 101-111.https://doi.org/10.1016/j.jgeb.2017.01.003
Brown, P.N., Lund, J.A., & Murch, S.J. (2017). A botanical, phytochemical and ethnomedicinal review of the genus Mitragyna korth: Implications for products sold as kratom. Journal of Ethnopharmacology, 202, 302-325. https://doi.org/10.1016/j.jep.2017.03.020
Chen, L., Cai, Y., Zhou, G., Shi, X., Su, J., Chen, G., & Lin, K. (2014). Rapid Sanger sequencing of the 16S rRNA gene for identification of some common pathogens. PLOS ONE, 9(2), e88886.https://doi.org/10.1371/journal.pone.0088886
Doilom, M., Guo, J.W., Phookamsak, R., Mortimer, P.E., Karunarathna, S.C., Dong, W., Liao, C.F., Yan, K., Pem, D., Suwannarach, N., Promputtha, I., Lumyong, S., & Xu, J.C. (2020). Screening of phosphate-solubilizing fungi from air and soil in Yunnan, China: four novel species in Aspergillus, Gongronella, Penicillium, and Talaromyces. Frontiers in Microbiology, 11, 585215. https://doi.org/10.3389/fmicb.2020.585215
Ercole, T.G., Liviero, R., Terra, L.A., Zocolo, G.J., Klepa, M.S., Ribeiro, R.A., Nogueira M.A., & Hungria, M. (2026). Integrated genome mining and phytohormone profiling of six plant growth-promoting elite bacterial strains. Archives of Microbiology 208, 152 https://doi.org/10.1007/s00203-025-04712-6
Harahap, R.T., Azizah, I.R., Setiawati, M.R., Herdiyantoro, D., & Simarmata, T. (2023). Enhancing upland rice growth and yield with indigenous plant growth-promoting rhizobacteria (PGPR) isolate at N-fertilizers dosage. Agriculture, 13(10), 1987. https://doi.org/10.3390/agriculture13101987
Jasim, B., Joseph, A.A., John, C.J., Mathew, J., & Radhakrishnan, E.K. (2014). Isolation and characterization of plant growth promoting endophytic bacteria from the rhizome of Zingiber officinale. 3 Biotech, 4(2), 197–204.https://doi.org/10.1007/s13205-013-0143-3
Jatav, P., Gupta, A., Ahirwar, S.S., Jatav, S., Jatav, A., & Kushwaha, K. (2017). Production plant growth hormones indole-3-acetic acid (IAA) using Bacillus by bath fermentation. Global Journal of Bioscience and Biotechnology, 6, 612-616.
Khan, A. L., Halo, B. A., Elyassi, A., Ali, S., Al-Hosni, K., Hussain, J., Al-Harrasi, A., & Lee, J. (2016). Indole acetic acid and ACC deaminase from endophytic bacteria improves the growth of Solanum lycopersicum. Electronic Journal of Biotechnology, 21(C), 58-64.https://doi.org/10.1016/j.ejbt.2016.02.001
Khan, M.A., Shahid, M., Celik, I., Khan, H.M., Shahzad, A., Husain, F.M., & Adil, M. (2024). Attenuation of quorum sensing regulated virulence functions and biofilm of pathogenic bacteria by medicinal plant Artemisia annua and its phytoconstituent 1, 8-cineole. Microscopy Research and Technique, 87(1):133-148. https://doi.org10.1002/jemt.24418.
Lin, Z., Qiao, Y., Ge, J., Lu, L., Xie, R., & Tian, S. (2025). Novel plant growth-promoting endophytic bacteria, Stenotrophomonas maltophilia SaRB5, facilitate phytoremediation by plant growth and cadmium absorption in Salix suchowensis. Ecotoxicology and Environmental Safety, 303. 118967. https://doi.org/10.1016/j.ecoenv.2025.113120
Lipková, N., Cinkocki, R., Maková, J., Medo, J., & Javoreková, S. (2021). Characterization of endophytic bacteria of the genus Bacillus and their influence on the growth of maize (Zea mays) in vivo. Journal of Microbiology, Biotechnology and Food Science, 10,1-5. https://doi.org/10.15414/jmbfs.3602
Liyuwardi, F. P. (2024). Antibacterial properties of endophytic bacteria isolated from Kalimantan's kratom (Mitragyna speciosa) [Undergraduate thesis]. Indonesia International Institute for Life Sciences Repository. https://repository.i3l.ac.id/jspui/handle/123456789/1131
Maharana, R., & Dhal, N.K. (2022). Solubilization of rock phosphate by phosphate solubilizing bacteria isolated from effluent treatment plant sludge of a fertilizer plant. Folia Microbiology, 67, 605–615. https://doi.org/10.1007/s12223-022-00953-w
Masrukhin, Putri AL, Sulistiyani TR, Ilyas, M., Purnaningsih, I., Saskiawan, I., & Niam, M.Y. (2021). Antifungal activity of bacterial isolates from straw mushroom cultivation medium against phytopathogenic fungi. Journal of Tropical Biodiversity and Biotechnology, 6, 1-9. https://doi.org/10.22146/jtbb.59235
Mir, M.I., Kumar, B.K., Gopalakrishnan, S., Vadlamudi, S., & Hameeda, B. (2021). Characterization of rhizobia isolated from leguminous plants and their impact on the growth of ICCV 2 variety of chickpea (Cicer arietinum L.). Heliyon, 7(11), e08321, 1-13. https://doi.org/10.1016/j.heliyon.2021.e08321
Moon, Y.S., & Ali, S. (2022). Possible mechanisms for the equilibrium of ACC and role of ACC deaminase-producing bacteria. Applied Microbiology and Biotechnology, 106: 877–887. https://doi.org/10.1007/s00253-022-11772-x
Naing, A.H., Maung, T.T., & Kim, C.K. (2021). The ACC deaminase-producing plant growth-promoting bacteria: Influences of bacterial strains and ACC deaminase activities in plant tolerance to abiotic stress. Physiology Plantarum, 173(4), 1992–2012. https://doi.org/10.1111/ppl.13545.
Nishihara, A., Thiel, V., Matsuura, K., McGlynn, S. E., & Haruta, S. (2018). Phylogenetic diversity of nitrogenase reductase genes and possible nitrogen-fixing bacteria in thermophilic chemosynthetic microbial communities in Nakabusa hot springs. Microbes and Environments, 33(4), 357–365.https://doi.org/10.1264/jsme2.ME18041.
Ntabo, R.M., Nyamache, A.K., Lwande, W., Kabii, J., & Nonoh, J. (2018). Enzymatic activity of endophytic bacterial isolates from selected mangrove plants in Kenya. The Open Microbiology Journal, 12, 354-363.
Pitiwittayakul, N., Niyomvong, N., Tedsree, N., Somphong, A., Chamroensaksri, N., Phongsopitanun, W., & Tanasupawat, S. (2026). Streptomyces mitragynae sp. nov., isolated from Mitragyna speciosaleaves in Thailand, with antimicrobial activity and secondary metabolite profiling. International Journal of Systematic and Evolutionary Microbiology 76(1).https://doi.org/10.1099/ijsem.0.007050
Ragot, S.A., Kertesz, M.A., & Bünemann, E.K. (2015). phoD alkaline phosphatase gene diversity in soil. Applied and Environmental Microbiology, 81(20), 7281–7289. https://doi.org/10.1128/AEM.01823-15
Rahman, L., Mukhtar, A., Ahmad, S., Rahman, L., Ali, M., Saeed, M., & Shinwari, Z.K. (2022). Endophytic bacteria of Fagonia indica Burm. F revealed to harbour rich secondary antibacterial metabolites. PLoS ONE 17:e0277825. https://doi.org/10.1371/journal.pone.0277825
Ramanuj, K.B., & Shelat, H.N. (2019). Plant growth promoting the potential of bacterial endophytes from medicinal plants. Advance Research, 13,1-15. https://doi.org/10.9734/AIR/2018/40014
Rana, K.L., Kour, D., Kaur, T., Negi, R., Devi1, R., Yadav, N., Rai, P.K., Singh, S., Rai, A.K., Yadav, A., Sayyed, R.Z., & Yadav, A.N. (2023). Endophytic nitrogen-fixing bacteria: untapped treasurer for agricultural sustainability. Journal of Applied Biology and Biotechnology, 11,75-93. https://doi.org/10.7324/JABB.2023.110207
Semenzato, G., & Fani, R. (2024). Endophytic bacteria: A sustainable strategy for enhancing medicinal plant cultivation and preserving microbial diversity. Frontiers in Microbiology, 15, Article 1477465. https://doi.org/10.3389/fmicb.2024.1477465
Sharma, M., Sood, G. & Chauhan, A. (2025). Bacterial endophytes of medicinal plants: Applications and recent developments. Current Microbiology, 82(11), 519. https://doi.org/10.1007/s00284-025-04499-1
Sharma, A., Singh, P., Sarmah, B.K., & Nandi, S.P. (2020). Isolation of cellulose-degrading endophyte from Capsicum chinense and determination of its cellulolytic potential. Biointerface Research in Applied Chemistry, 10, 6964 – 6973. https://doi.org/10.33263/BRIAC106.69646973
Sulistiyani, T.R., & Meliah, S. (2017). Isolation and characterization of nitrogen fixing endophytic bacteria associated with sweet sorghum (Sorghum bicolor). Proceedia, 110-117.
Sepp, S. K., Vasar, M., Davison, J., Oja, J., Anslan, S., Al-Quraishy, S., Bahram, M., Bueno, C. G., Cantero, J. J., Fabiano, E. C., Decocq, G., Drenkhan, R., Fraser, L., Garibay Oriel, R., Hiiesalu, I., Koorem, K., Kõljalg, U., Moora, M., Mucina, L., Opik, M., Polme, S., Partel, M., Phosri, C., Semchenko, M., Vahter, T., Vasco Palacios, A.M., Tedersoo, L., & Zobel, M. (2023). Global diversity and distribution of nitrogen-fixing bacteria in the soil. Frontiers in Plant Science, 14, 1100235. https://doi.org/10.3389/fpls.2023.1100235
Tang, A., Haruna, A.O., Majid, N.M.A., & Jalloh, M.B. (2020). Potential PGPR properties of cellulolytic, nitrogen-fixing, phosphate-solubilizing bacteria in rehabilitated tropical forest soil. Microorganisms, 8(3), 442. https://doi.org/10.3390/microorganisms8030442
Tariq, A., & Ahmed, A. (2022). Phosphate-solubilizing rhizobacteria as sustainable management strategy in agrobiology. IntechOpen. https://doi.org/10.5772/intechopen.108657
Tsipinana, S., Obi, L., Amoo, S., & Adeleke, R. (2024) Plant growth-promoting potential of bacterial endophytes isolated from Lessertia frutescens. South African Journal of Botany,174,768-778. https://doi.org/10.1016/j.sajb.2024.09.043.
Venkatramanan, M., & Nalini, E. (2024). Regulation of virulence in Chromobacterium violaceum and strategies to combat it. Frontiers in Microbiology, 15, 1303595. https://doi.org/10.3389/fmicb.2024.1303595
Wu, T., Xu, J., Liu, J., Liu, Y., Li, Y., Yu, X., Zhao, K., Gu, Y., Xu, K., Chen, C., & Chen, Q. (2019). Characterization and initial application of endophytic Bacillus safensis strain zy16 for improving phytoremediation of oil-contaminated saline soils. Frontiers in Microbiology, 10, 1-9.https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2019.00991/full
Xu, L., Cao, H., Li, C., Wang, C., He, N., Hu, S., Yao, M., Wang, C., Wang, J., Zhou, S., & Li, X. (2021). The importance of rare versus abundant phoD-harboring subcommunities in driving soil alkaline phosphatase activity and available P content in Chinese steppe ecosystems. Soil Biology and Biochemistry, 164, 108491.https://doi.org/10.1016/j.soilbio.2021.108491
Zaghloul, R.A., Abou-Aly, H.E., Tewfike, T.A., & Ashry, N.M. (2016). Isolation and characterization of endophytic bacteria isolated from legumes and non-legume plants. Egyptian Journal of Pure Applied Microbiology, 10, 277–290. https://doi.org/10.13140/RG.2.1.3653.0325
Zhang, Y., Kang, X., Liu, H., Liu, Y., Li, Y., Yu, X., Zhao, K., Gu, Y., Xu, K., Chen, C., & Chen, Q. (2018). Endophytes isolated from ginger rhizome exhibit growth-promoting potential for Zea mays. Archives of Agronomy and Soil Sciences, 64, 1302-1314. https://doi.org/10.1080/03650340.2018.1430892
Zhang, X., Tong, J., Dong, M., Akhtar, K., & He, B. (2022). Isolation, identification and characterization of nitrogen-fixing endophytic bacteria and their effects on cassava production. PeerJ, 10, e12677.https://doi.org/10.7717/peerj.12677
Zhang, M., Lyndon, A., Kanumuri, S. R. R., Sharma, A., Pearson, B. J., McCurdy, C. R., & Chen, J. (2025). Alkaloid biosynthesis in medicinal crop kratom (Mitragyna speciosa) varies with postharvest, genetic, and seasonal factors. Frontiers in Plant Science, 16, Article 1653916. https://doi.org/10.3389/fpls.2025.1653916
Zhao, L., Xu, Y., Lai, X.H., Shan, C., Deng, Z., & Ji, Y. (2015). Screening and characterization of endophytic Bacillus and Paenibacillus strains from medicinal plant Lonicera japonica for use as potential plant growth promoters. Brazilian Journal of Microbiology, 46(4), 977–989. https://doi.org/10.1590/s1517-838246420140024
Zhao, Y., Ding, Y., Xu, L., & Sun, J.Q. (2024). A comprehensive comparative genomic analysis revealed that plant growth promoting traits are ubiquitous in strains of Stenotrophomonas. Frontiers in Microbiology, 15, Article 1395477. https://doi.org/10.3389/fmicb.2024.1395477
strains during tomato plant infection. Microbial Biotechnology, 4(1), 82–88. https://doi.org/10.1111/j.1751-7915.2010.00214.x
Downloads
Publicado
Como Citar
Edição
Seção
Licença
Copyright (c) 2026 Scientia Agropecuaria

Este trabalho está licenciado sob uma licença Creative Commons Attribution-NonCommercial 4.0 International License.
Los autores que publican en esta revista aceptan los siguientes términos:
a. Los autores conservan los derechos de autor y conceden a la revista el derecho publicación, simultáneamente licenciada bajo una licencia de Creative Commons que permite a otros compartir el trabajo, pero citando la publicación inicial en esta revista.
b. Los autores pueden celebrar acuerdos contractuales adicionales separados para la distribución no exclusiva de la versión publicada de la obra de la revista (por ejemplo, publicarla en un repositorio institucional o publicarla en un libro), pero citando la publicación inicial en esta revista.
c. Se permite y anima a los autores a publicar su trabajo en línea (por ejemplo, en repositorios institucionales o en su sitio web) antes y durante el proceso de presentación, ya que puede conducir a intercambios productivos, así como una mayor citación del trabajo publicado (ver efecto del acceso abierto).




