Porcine reproductive physiology
DOI:
https://doi.org/10.17268/sci.agropecu.2026.030Keywords:
swine, reproduction, performance, efficiency, developmentAbstract
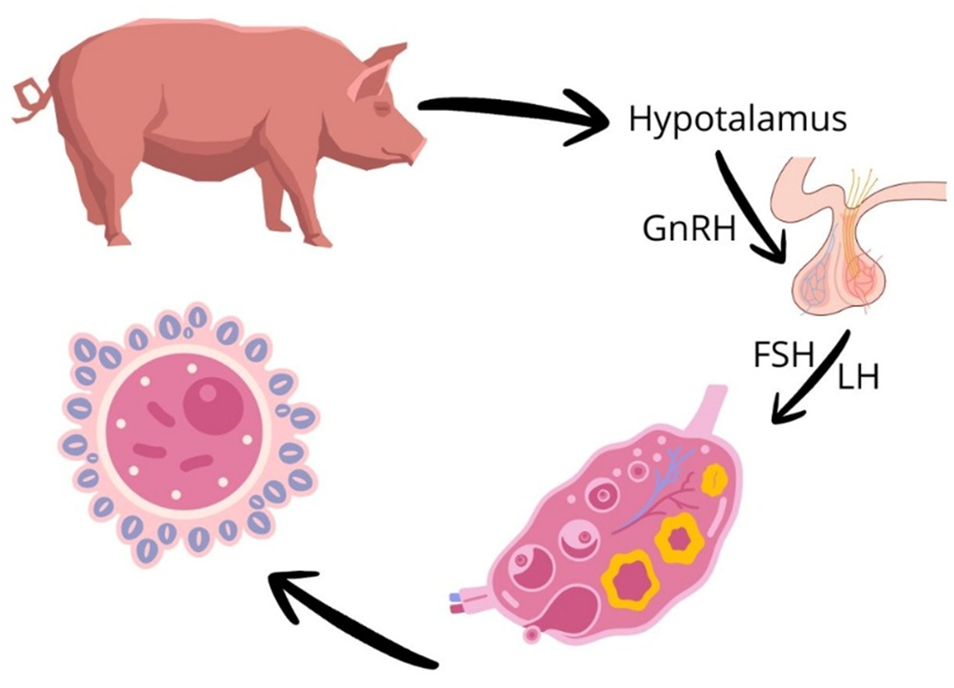
The objective of this review is to synthesize current scientific knowledge on the physiological processes that govern porcine reproduction and their interactions with key metabolic and hormonal factors. It addresses gametogenesis (oogenesis and spermatogenesis), the estrous cycle, folliculogenesis, and the hypothalamic-pituitary-gonadal (HPG) axis, as well as critical fertilization events such as sperm capacitation, the acrosomal reaction, and fertilization itself. Embryogenesis and gestation are also examined, with emphasis on underlying biochemical and hormonal mechanisms. Special attention is given to metabolic analytes—glucose, triglycerides, and cholesterol—and their influence on the synthesis and regulation of reproductive hormones including estradiol (E2), progesterone (P4), and testosterone. The review focuses on physiological and biochemical aspects specific to domestic pigs (Sus scrofa domesticus), offering a targeted analysis relevant to both livestock production and biomedical research. The integration of metabolic and endocrine perspectives enhances our understanding of how nutrient availability, energy balance, and environmental stress affect reproductive efficiency. Key findings highlight the central role of the HPG axis in coordinating gonadotropin release and steroidogenesis, the impact of metabolic homeostasis on gamete quality and embryo viability, and the importance of hormonal feedback in regulating the reproductive cycle. Practical applications such as estrus synchronization and artificial insemination are discussed in light of these physiological insights. In conclusion, porcine reproductive physiology is shaped by a complex interplay of hormonal and metabolic factors that determine fertility and gestational success. Despite recent advances, further research is needed to better understand metabolic–hormonal crosstalk, particularly during gestation, and to enhance outcomes in both agricultural and biomedical contexts.
References
Ackerman, D. (2022). 2022 African swine fever virus research review (L. Robinson, Ed.). STAR-IDAZ International Research Consortium.
Ake, A. S., Ayo, J. O., Aluwong, T., Mohammed, A., & Minka, N. S. (2023). Melatonin modulates rectal and body surface temperatures and their circadian rhythmicity in donkeys (Equus asinus) subjected to packing during the hot-dry season. International Journal of Biometeorology, 67(2), 389-404. https://doi.org/10.1007/s00484-022-02418-8
Akhavizadegan, H., Farsani, R. M., Golmohammadi, P., & Van Der Walt, S. (2025). Chapter 11 - Infertility and the chromosomal abnormalities. In S. M. Kazem Aghamir (Ed.), Genetics and Epigenetics of Genitourinary Diseases (pp. 151-180). Academic Press. https://doi.org/10.1016/B978-0-443-27302-5.00004-8
Albrizio, M., Lacalandra, G. M., & Cinone, M. (2022). The role of bicarbonate in the modulation of capacitation, spontaneous acrosome reaction and motility of equine fresh and frozen spermatozoa. Theriogenology, 187, 112-118. https://doi.org/10.1016/j.theriogenology.2022.04.032
Amini, M., & Benson, J. D. (2023). Technologies for vitrification based cryopreservation. Bioengineering, 10(5), 508. https://doi.org/10.3390/bioengineering10050508
Aponte, P. M., Gutierrez-Reinoso, M. A., & Garcia-Herreros, M. (2024). Bridging the Gap: Animal Models in Next-Generation Reproductive Technologies for Male Fertility Preservation. Life, 14(1), 17. https://doi.org/10.3390/life14010017
Arboccó, F. C. V., Persia, F. A., Zyla, L., Bernal, N., Sasso, V. C., Santiano, F., Gomez, S., Bruna, F., Pistone-Creydt, V., Lopez-Fontana, C., Jahn, G. A., Hapon, M. B., & Carón, R. W. (2023). Long-term impact of hypothyroidism during gestation and lactation on the mammary gland. Journal of Developmental Origins of Health and Disease, 14(1), 122-131. https://doi.org/10.1017/S2040174422000320
Báez, F., López Darriulat, R., Rodríguez-Osorio, N., & Viñoles, C. (2022). Effect of season on germinal vesicle stage, quality, and subsequent in vitro developmental competence in bovine cumulus-oocyte complexes. Journal of Thermal Biology, 103, 103171. https://doi.org/10.1016/j.jtherbio.2021.103171
Bai, L., Zhao, Y., Zhou, Y., Song, Y., Xiao, H., Zhao, G., Wang, Z., & Li, X. (2025). Advances in immunological sorting of X and Y chromosome-bearing sperm: from proteome to sex-specific proteins. Frontiers in Veterinary Science, 12. https://doi.org/10.3389/fvets.2025.1523491
Ballester, M., Amills, M., González-Rodríguez, O., Cardoso, T. F., Pascual, M., González-Prendes, R., Panella-Riera, N., Díaz, I., Tibau, J., & Quintanilla, R. (2018). Role of AMPK signalling pathway during compensatory growth in pigs. BMC Genomics, 19(1), 682. https://doi.org/10.1186/s12864-018-5071-5
Balu, R., Ramachandran, S. S., Mathimaran, A., Jeyaraman, J., & Paramasivam, S. G. (2022). Functional significance of mouse seminal vesicle sulfhydryl oxidase on sperm capacitation in vitro. Molecular Human Reproduction, 28(9). https://doi.org/10.1093/molehr/gaac025
Bao, J., & Bedford, M. T. (2016). Epigenetic regulation of the histone-to-protamine transition during spermiogenesis. Reproduction, 151(5), R55-R70. https://doi.org/10.1530/REP-15-0562
Baranizadeh, K., Mahboobian, M. M., Amiri, I., Tavilani, H., & Shafiee, G. (2022). Effects of progesterone nanoparticles on the sperm capacitation and acrosome reaction in asthenozoospermia men. Andrologia, 54(1), e14258. https://doi.org/10.1111/and.14258
Bee, G., Quiniou, N., Maribo, H., Zamaratskaia, G., & Lawlor, P. G. (2020). Strategies to meet nutritional requirements and reduce boar taint in meat from entire male pigs and immunocastrates. Animals, 10(11), 1950. https://doi.org/10.3390/ani10111950
Bortolozzo, F. P., Zanin, G. P., Christ, T. S., Rech, R. D., da Rosa Ulguim, R., & Mellagi, A. P. G. (2024). Artificial insemination and optimization of the use of seminal doses in swine. Animal Reproduction Science, 269, 107501. https://doi.org/10.1016/j.anireprosci.2024.107501
Bortolozzo, F. P., Zanin, G. P., Ulguim, R. d. R., & Mellagi, A. P. G. (2023). Managing reproduction in hyperprolific sow herds. Animals, 13(11), 1842. https://doi.org/10.3390/ani13111842
Bremer, A. A., & Miller, W. L. (2014). Chapter 13 - Regulation of Steroidogenesis. In A. Ulloa-Aguirre and P. M. Conn (Eds.), Cellular Endocrinology in Health and Disease (pp. 207-227). Academic Press. https://doi.org/10.1016/B978-0-12-408134-5.00013-5
Cañón-Beltrán, K., Cajas, Y. N., González, E., Fernández-González, R., Fierro, N., Lorenzo, P. L., Arias-Álvarez, M., García-García, R. M., Gutiérrez-Adán, A., & Rizos, D. (2023). Acquisition of fertilization competence in guinea pig spermatozoa under different capacitation protocols. Theriogenology, 198, 231-240. https://doi.org/10.1016/j.theriogenology.2022.12.042
Casarini, L., & Crépieux, P. (2019). Molecular mechanisms of action of FSH. Frontiers in Endocrinology, 10, 305. https://doi.org/10.3389/fendo.2019.00305
Chen, P. R., Redel, B. K., Kerns, K. C., Spate, L. D., & Prather, R. S. (2021). Challenges and considerations during in vitro production of porcine embryos. Cells, 10(10), 2770. https://doi.org/10.3390/cells10102770
Chen, Y., Wang, S., & Zhang, C. (2025). The differentiation fate of granulosa cells and the regulatory mechanism in ovary. Reproductive Sciences, 32(5), 1414-1426. https://doi.org/10.1007/s43032-024-01682-w
Choe, Y.-h., Sorensen, J., Garry, D. J., & Garry, M. G. (2022). Blastocyst complementation and interspecies chimeras in gene edited pigs. Frontiers in Cell and Developmental Biology, 10. https://doi.org/10.3389/fcell.2022.1065536
Chou, J.-Y., & Parsons, T. D. (2022). A systematic review of the impact of housing on sow welfare during post-weaning and early pregnancy periods. Frontiers in Veterinary Science, 9, 903822. https://doi.org/10.3389/fvets.2022.903822
Choudhary, S., Kamboj, M. L., Ungerfeld, R., & Singh, P. (2022). Calf-cow and bull-cow management in buffaloes: Effects on growth, productive and reproductive performance of mothers and their calves. Reproduction in Domestic Animals, 57(11), 1428-1439. https://doi.org/10.1111/rda.14219
Contreras, M. J., Núñez-Montero, K., Bruna, P., García, M., Leal, K., Barrientos, L., & Weber, H. (2022). Bacteria and boar semen storage: Progress and challenges. Antibiotics, 11(12), 1796. https://doi.org/10.3390/antibiotics11121796
Dalen, K. T., & Li, Y. (2024). Chapter 3 - Regulation of lipid droplets and cholesterol metabolism in adrenal cortical cells. In G. Litwack (Ed.), Vitamins and Hormones (Vol. 124, pp. 79-136). Academic Press. https://doi.org/10.1016/bs.vh.2023.06.007
Das, P. K., Mukherjee, J., & Banerjee, D. (2023a). Female Reproductive Physiology. In P. K. Das, V. Sejian, J. Mukherjee, and D. Banerjee (Eds.), Textbook of Veterinary Physiology (pp. 513-568). Springer Nature Singapore. https://doi.org/10.1007/978-981-19-9410-4_22
Das, P. K., Mukherjee, J., & Banerjee, D. (2023b). Fertilisation, gestation and parturition. In P. K. Das, V. Sejian, J. Mukherjee, and D. Banerjee (Eds.), Textbook of Veterinary Physiology (pp. 569-613). Springer Nature Singapore. https://doi.org/10.1007/978-981-19-9410-4_23
Das, P. K., Mukherjee, J., & Banerjee, D. (2023c). Functional Morphology of the Male Reproductive System. In P. K. Das, V. Sejian, J. Mukherjee, and D. Banerjee (Eds.), Textbook of Veterinary Physiology (pp. 441-476). Springer Nature Singapore. https://doi.org/10.1007/978-981-19-9410-4_19
De Rensis, F., Saleri, R., Tummaruk, P., Techakumphu, M., & Kirkwood, R. N. (2012). Prostaglandin F2α and control of reproduction in female swine: a review. Theriogenology, 77(1), 1-11. https://doi.org/10.1016/j.theriogenology.2011.07.035
de Souza, V. G., Bandeira, L. B., Souza, N. C. S. e., Taboga, S. R., Martins, T. M. M., & Silva Perez, A. P. (2022). Prenatal and pubertal exposure to 17α-ethinylestradiol disrupts folliculo-genesis and promotes morphophysiological changes in ovaries of old gerbils (Meriones unguiculatus). Journal of Developmental Origins of Health and Disease, 13(1), 49-60. https://doi.org/10.1017/S2040174421000040
Deryabin, P. I., & Borodkina, A. V. (2024). The role of the endometrium in implantation: a modern view. International Journal of Molecular Sciences, 25(17), 9746. https://doi.org/10.3390/ijms25179746
Esbenshade, K. L., Ziecik, A. J., & Britt, J. H. (1990). Regulation and action of gonadotrophins in pigs. Journal of Reproduction and Fertility Supplement, 40, 19-32.
Fabová, Z., Loncová, B., & Sirotkin, A. V. (2023). MicroRNA miR-125b can suppress ovarian granulosa cell functions: Interrelationships with FSH. Cell Biochemistry & Function, 41(2), 177-188. https://doi.org/10.1002/cbf.3771
Faheem, M. S., Dessouki, S. M., Abdel- Rahman, F. E. S., & Ghanem, N. (2021). Physiological and molecular aspects of heat-treated cultured granulosa cells of Egyptian buffalo (Bubalus bubalis). Animal Reproduction Science, 224, 106665. https://doi.org/10.1016/j.anireprosci.2020.106665
Fang, W., Wen, X., Meng, Q., Liu, L., Xie, J., Zhang, H., & Everaert, N. (2020). Running head: heat affects cholesterol and bile acid alterations in cholesterol and bile acids metabolism in large white pigs during short-term heat exposure. Animals, 10(2), 359. https://doi.org/10.3390/ani10020359
Fidarov, A. F., Vihma, V., Bogautdinov, R. P., Morozkina, S. N., Shavva, A. G., & Tikkanen, M. J. (2015). Novel structural features increase the antioxidant effect of estrogen analogues on low density lipoprotein. The Journal of Steroid Biochemistry and Molecular Biology, 154, 142-149. https://doi.org/10.1016/j.jsbmb.2015.08.001
Flowers, W. L. (2022). Factors affecting the production of quality ejaculates from boars. Animal Reproduction Science, 246, 106840. https://doi.org/10.1016/j.anireprosci.2021.106840
Ford, E. A., Frost, E. R., Beckett, E. L., Roman, S. D., McLaughlin, E. A., & Sutherland, J. M. (2021). Transcriptomic profiling of neonatal mouse granulosa cells reveals new insights into primordial follicle activation. Biology of Reproduction, 106(3), 503-514. https://doi.org/10.1093/biolre/ioab193
Fuentes, N., & Silveyra, P. (2019). Chapter 3 - Estrogen receptor signaling mechanisms. In R. Donev (Ed.), Advances in Protein Chemistry & Structural Biology (Vol. 116, pp. 135-170). Academic Press. https://doi.org/10.1016/bs.apcsb.2019.01.001
Fujihara, Y., Kobayashi, K., Abbasi, F., Endo, T., Yu, Z., Ikawa, M., & Matzuk, M. M. (2023). PDCL2 is essential for sperm acrosome formation and male fertility in mice. Andrology, 11(5), 789-798. https://doi.org/10.1111/andr.13329
Galano, M., Venugopal, S., & Papadopoulos, V. (2022). Role of star and SCP2/SCPx in the transport of cholesterol and other lipids. International Journal of Molecular Sciences, 23(20), 12115. https://doi.org/10.3390/ijms232012115
Gałęska, E., Wrzecińska, M., Kowalczyk, A., & Araujo, J. P. (2022). Reproductive consequences of electrolyte disturbances in domestic animals. Biology, 11(7), 1006. https://doi.org/10.3390/biology11071006
Gao, D.-D., Lan, C.-F., Cao, X.-N., Chen, L., Lei, T.-L., Peng, L., Xu, J.-W., Qiu, Z.-E., Wang, L.-L., Sun, Q., Huang, Z.-Y., Zhu, Y.-X., Zhou, W.-L., & Zhang, Y.-L. (2022). G protein-coupled estrogen receptor promotes acrosome reaction via regulation of Ca2+ signaling in mouse sperm. Biology of Reproduction, 107(4), 1026-1034. https://doi.org/10.1093/biolre/ioac136
Gardela, J., Ruiz-Conca, M., Wright, D., López-Béjar, M., Martínez, C. A., Rodríguez-Martínez, H., & Álvarez-Rodríguez, M. (2022). Semen modulates cell proliferation and differentiation-related transcripts in the pig peri-ovulatory endometrium. Biology, 11(4), 616. https://doi.org/10.3390/biology11040616
Ghezelayagh, Z., Khoshdel-Rad, N., & Ebrahimi, B. (2022). Human ovarian tissue in-vitro culture: primordial follicle activation as a new strategy for female fertility preservation. Cytotechnology, 74(1), 1-15. https://doi.org/10.1007/s10616-021-00510-2
Giersch, G. E. W., Charkoudian, N., Morrissey, M. C., Butler, C. R., Colburn, A. T., Caldwell, A. R., Kavouras, S. A., & Casa, D. J. (2022). Corrigendum: estrogen to progesterone ratio and fluid regulatory responses to varying degrees and methods of dehydration. Frontiers in Sports and Active Living, 4. https://doi.org/10.3389/fspor.2022.848595
Gil, M. A., Parrilla, I., Cuello, C., & Martinez, E. A. (2025). Current status of nonsurgical embryo transfer in swine. Reproduction, Fertility and Development, 37(1), RD24137. https://doi.org/10.1071/RD24137
Glencorse, D., Grupen, C. G., & Bathgate, R. (2025). A Review of the Monitoring Techniques Used to Detect Oestrus in Sows. Animals, 15(3), 331. https://doi.org/10.3390/ani15030331
Goszczynski, D. E., Tinetti, P. S., Choi, Y. H., Hinrichs, K., & Ross, P. J. (2021). Genome activation in equine in vitro–produced embryos. Biology of Reproduction, 106(1), 66-82. https://doi.org/10.1093/biolre/ioab173
Guarnotta, V., Amodei, R., Frasca, F., Aversa, A., & Giordano, C. (2022). Impact of chemical endocrine disruptors and hormone modulators on the endocrine system. International Journal of Molecular Sciences, 23(10), 5710. https://doi.org/10.3390/ijms23105710
Guo, S., Liu, C., Wang, Y., Chen, F., Zhu, J., Li, S., & Li, E. (2024). Effect of resveratrol on spermatogenesis in breeding boars and the proteomic analysis for testes. Reproductive Biology, 24(4), 100930. https://doi.org/10.1016/j.repbio.2024.100930
Harris, R. B. S. (2014). Direct and indirect effects of leptin on adipocyte metabolism. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease, 1842(3), 414-423. https://doi.org/10.1016/j.bbadis.2013.05.009
Heinlein, C. A., & Chang, C. (2002). Theroles of androgen receptors and androgen-binding proteins in nongenomic androgen actions. Molecular Endocrinology, 16(10), 2181-2187. https://doi.org/10.1210/me.2002-0070
Heringstad, B., & Wethal, K. B. (2023). Cow activity measurements can be used to define new fertility traits for use in genetic evaluation. JDS Communications, 4(2), 99-100. https://doi.org/10.3168/jdsc.2022-0251
Hosseinzadeh, S., & Masoudi, A. A. (2024). Investigating the expression of fertility-regulating LncRNAs in multiparous and uniparous Shal ewe's ovaries. Genome, 67(3), 78-89. https://doi.org/10.1139/gen-2023-0055
Howard, H. J., & Ford, J. J. (1994). Differential steroidogenic response of subpopulations of porcine granulosa cells to insulin-like growth factor-1 (IGF-1) or IGF-1 analogs. Biology of Reproduction, 51(1), 108-115. https://doi.org/10.1095/biolreprod51.1.108
Hu, Y., Li, W., Cheng, X., Yang, H., She, Z.-G., Cai, J., Li, H., & Zhang, X.-J. (2024). Emerging roles and therapeutic applications of arachidonic acid pathways in cardiometabolic diseases. Circulation Research, 135(1), 222-260. https://doi.org/10.1161/CIRCRESAHA.124.324383
Hu, Z., Yu, Y., Huang, G., Li, J., Yang, C., Long, A., Tang, J., Che, T., Zhao, S., & Zhang, T. (2026). IGF‐1 deficiency serves as an integrated biomarker pathogenic driver and predictor in poor ovarian response. Advanced Science, e14483. https://doi.org/10.1002/advs.202514483
Hunter, R. H. F. (2012). Components of oviduct physiology in eutherian mammals. Biological Reviews, 87(1), 244-255. https://doi.org/10.1111/j.1469-185X.2011.00196.x
Jackson, L. A., Shi, H., Acevedo, J., Lee, S., Annabi, N., Word, R. A., & Florian-Rodriguez, M. (2022). Effect of gelatin methacryloyl hydrogel on healing of the guinea pig vaginal wall with or without mesh augmentation. International Urogynecology Journal, 33(8), 2223-2232. https://doi.org/10.1007/s00192-021-05031-2
Jia, H.-H., Xu, Y.-T., Yin, Z.-P., Wu, X.-M., Qing, M., Fan, Y.-J., Song, X., Xie, K.-D., Xie, Z.-Z., Xu, Q., Deng, X.-X., & Guo, W.-W. (2021). Transcriptomes and DNA methylomes in apomictic cells delineate nucellar embryogenesis initiation in citrus. DNA Research, 28(5), dsab014. https://doi.org/10.1093/dnares/dsab014
Jochems, R., Gaustad, A. H., Styrishave, B., Zak, L. J., Oskam, I. C., Grindflek, E., Myromslien, F. D., Kommisrud, E., & Krogenæs, A. K. (2022). Follicular fluid steroid hormones and in vitro embryo development in Duroc and Landrace pigs. Theriogenology, 190, 15-21. https://doi.org/10.1016/j.theriogenology.2022.07.004
Kaiser, U. B. (2011). Chapter 7 - Gonadotropin hormones. In S. Melmed (Ed.), The Pituitary (Third Edition) (pp. 205-260). Academic Press. https://doi.org/10.1016/B978-0-12-380926-1.10007-0
Kang, H.-I., Lee, C.-B., Kwon, S.-H., Park, J.-M., Kang, K.-S., and Shim, D. (2021a). Comparative transcriptome analysis during developmental stages of direct somatic embryogenesis in Tilia amurensis Rupr. Scientific Reports, 11(1), 6359. https://doi.org/10.1038/s41598-021-85886-z
Kang, Y., Li, W., Zhang, L., & Qi, L. (2021b). Over-expression of the Cell-Cycle gene LaCDKB1;2 promotes cell proliferation and the formation of normal cotyledonary embryos during Larix kaempferi somatic embryogenesis. Genes, 12(9), 1435. https://doi.org/10.3390/genes12091435
Kang, Y. C., Wetterer, R. T., Karimov, R. R., Kojima, M., Surke, M., Martín-Torres, I., Nicolai, J., Elkin, M., & Hartwig, J. F. (2024). Substitution, elimination, and integration of methyl groups in terpenes initiated by C–H bond functionalization. ACS Central Science, 10(11), 2016-2027. https://doi.org/10.1021/acscentsci.4c01108
Karzan Jalal, S. (2021). The major pathways of lipids (triglyceride and cholesterol) and lipoprotein metabolism. Zanco Journal of Pure and Applied Sciences, 33(4), 61-72. https://doi.org/10.21271/ZJPAS.33.4.6
Katzenellenbogen, B. S. (2000). Mechanisms of action and cross-talk between estrogen receptor and progesterone receptor pathways. Journal of the Society for Gynecologic Investigation, 7(1_suppl), S33-S37. https://doi.org/10.1177/1071557600007001s10
Kawashima, N., Tokuda, J., Yagi, T., & Takahashi, K. (2022). Isolation of a Nocardiopsis chromatogenes strain that degrades PLA (polylactic acid) in pig waste-based compost. Archives of Microbiology, 204(10), 599. https://doi.org/10.1007/s00203-022-03144-w
Kern, C., Wu, W., Lu, C., Zhang, J., Zhao, Y., Ocon-Grove, O. M., Sutovsky, P., Diaz, F., & Liu, W.-S. (2023). Role of the bovine PRAMEY protein in sperm function during in vitro fertilization (IVF). Cell and Tissue Research, 391(3), 577-594. https://doi.org/10.1007/s00441-022-03717-7
Knox, R. V. (2019). Physiology and endocrinology symposium: factors influencing follicle development in gilts and sows and management strategies used to regulate growth for control of estrus and ovulation. Journal of Animal Science, 97(4), 1433-1445. https://doi.org/10.1093/jas/skz036
Koketsu, Y., Tani, S., & Iida, R. (2017). Factors for improving reproductive performance of sows and herd productivity in commercial breeding herds. Porcine Health Management, 3(1), 1. https://doi.org/10.1186/s40813-016-0049-7
Kolomeitseva, G. L., Babosha, A. V., Ryabchenko, A. S., & Tsavkelova, E. A. (2021). Megasporogenesis, megagametogenesis, and embryogenesis in Dendrobium nobile (Orchidaceae). Protoplasma, 258(2), 301-317. https://doi.org/10.1007/s00709-020-01573-2
Kozieradzka-Kiszkurno, M. (2021). Embryogenesis in Crassulaceae: structural aspect of suspensor development. Postepy Biochemii, 68(1), 38-45. https://doi.org/10.18388/pb.2021_406
Kraeling, R. R., & Webel, S. K. (2015). Current strategies for reproductive management of gilts and sows in North America. Journal of Animal Science and Biotechnology, 6(1), 3. https://doi.org/10.1186/2049-1891-6-3
Krajnik, K., Mietkiewska, K., Skowronska, A., Kordowitzki, P., & Skowronski, M. T. (2023). Oogenesis in women: from molecular regulatory pathways and maternal age to stem cells. International Journal of Molecular Sciences, 24(7), 6837. https://doi.org/10.3390/ijms24076837
Kubikova, N., Keefe, D. L., Wells, D., Oktay, K. H., & Feinberg, E. C. (2023). Should we use CRISPR gene editing in human embryos? Fertility and Sterility, 120(4), 737-744. https://doi.org/10.1016/j.fertnstert.2023.08.009
Kumar, D., Kumar, V., & Jalwal, P. (2016). Systemic nomenclature of steroids [cyclopentaphenanthrene ring] their derivatives-an overview. International Journal of Chemical Studies, 4(3), 124-132.
Kurata, S., Umezu, K., Takamori, H., Hiradate, Y., Hara, K., & Tanemura, K. (2022). Exogenous gamma-aminobutyric acid addition enhances porcine sperm acrosome reaction. Animal Science Journal, 93(1), e13744. https://doi.org/10.1111/asj.13744
Lange-Consiglio, A., Capra, E., Giuliani, D., Canesi, S., Funghi, F., Bosi, G., Cretich, M., Frigerio, R., Galbiati, V., & Cremonesi, F. (2022). Endometrial and oviduct extra-cellular vescicles for in vitro equine sperm hyperactivation and oocyte fertilization. Theriogenology, 194, 35-45. https://doi.org/10.1016/j.theriogenology.2022.09.023
Laporta, J., Dado-Senn, B., & Skibiel, A. L. (2022). Late gestation hyperthermia: epigenetic programming of daughter's mammary development and function. Domestic Animal Endocrinology, 78, 106681. https://doi.org/10.1016/j.domaniend.2021.106681
Lema, S. C., Chow, M. I., Dittman, A. H., May, D., & Housh, M. J. (2022). Accustomed to the heat: Temperature and thyroid hormone influences on oogenesis and gonadal steroidogenesis pathways vary among populations of Amargosa pupfish (Cyprinodon nevadensis amargosae). Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology, 272, 111280. https://doi.org/10.1016/j.cbpa.2022.111280
Lei, Y., Liu, H., Xiang, Q., Liu, Y., Wu, D., Zhang, J., & Lin, Y. (2026). Dietary lycopene mitigates reproductive impairment in heat-stressed Rongchang boars: roles of antioxidant, anti-inflammatory and Nrf2 pathway. Antioxidants, 15(2), 245. https://doi.org/10.3390/antiox15020245
Li, J., Yang, H., Qin, K., Wei, L., Xia, X., Zhu, F., Tan, X., Xue, C., & Zhao, Q. (2022a). Effect of pig manure-derived sulfadiazine on species distribution and bioactivities of soil ammonia-oxidizing microorganisms after fertilization. Journal of Hazardous Materials, 423, 126994. https://doi.org/10.1016/j.jhazmat.2021.126994
Li, S., Wang, J., Li, J., Yue, M., Liu, C., Ma, L., & Liu, Y. (2022b). Integrative analysis of transcriptome complexity in pig granulosa cells by long-read isoform sequencing. PeerJ, 10, e13446. https://doi.org/10.7717/peerj.13446
Li, W., Ye, C., He, M., Ko, W. K. W., Cheng, C. H. K., Chan, Y. W., & Wong, A. O. L. (2024). Differential involvement of cAMP/PKA-, PLC/PKC- and Ca2+/calmodulin-dependent pathways in GnRH-induced prolactin secretion and gene expression in grass carp pituitary cells. Frontiers in Endocrinology, 15. https://doi.org/10.3389/fendo.2024.1399274
López-Gatius, F., Saleri, R., De Rensis, F., Llobera-Balcells, M., & Garcia-Ispierto, I. (2022). Transfer of a single fresh in vitro-produced embryo may prevent twin pregnancy without compromising the fertility of the cow. Reproduction in Domestic Animals, 57(4), 450-455. https://doi.org/10.1111/rda.14079
Lounsbury, K. (2009). Chapter 6 - Signal Transduction and Second Messengers. In M. Hacker, W. Messer, and K. Bachmann (Eds.), Pharmacology (pp. 103-112). Academic Press. https://doi.org/10.1016/B978-0-12-369521-5.00006-3
Lucy, M. C., & Safranski, T. (2017). Heat stress in pregnant sows: thermal responses and subsequent performance of sows and their offspring. Molecular Reproduction Development, 84(9), 946-956. https://doi.org/10.1002/mrd.22844
Lunney, J. K., Fang, Y., Ladinig, A., Chen, N., Li, Y., Rowland, B., & Renukaradhya, G. J. (2016). Porcine reproductive and respiratory syndrome virus (PRRSV): pathogenesis and interaction with the immune system. Annual Review of Animal Biosciences, 4(4), 129-154. https://doi.org/10.1146/annurev-animal-022114-111025
Ma, J., Xie, Q., Zhang, Y., Xiao, Q., Liu, X., Qiao, C., and Tian, Y. (2024). Advances in microfluidic technology for sperm screening and in vitro fertilization. Analytical and Bioanalytical Chemistry, 416(16), 3717-3735. https://doi.org/10.1007/s00216-023-05120-9
Maňásková, P., Pěknicová, J., Elzeinova, F., Ticha, M., & Jonakova, V. (2007). Origin, localization and binding abilities of boar DQH sperm surface protein tested by specific monoclonal antibodies. Journal of Reproductive Immunology, 74(1-2), 103-113. https://doi.org/10.1016/j.jri.2006.11.003
Mao, H., Chen, L., Bao, R., Weng, S., Wang, M., Xu, N., Qi, L., & Wang, J. (2022). Mechanisms of oogenesis-related long non-coding rnas in porcine ovaries treated with recombinant pig follicle-stimulating hormone. Frontiers in Veterinary Science, 8. https://doi.org/10.3389/fvets.2021.838703
Marchais, M., Gilbert, I., Bastien, A., Macaulay, A., & Robert, C. (2022). Mammalian cumulus-oocyte complex communication: a dialog through long and short distance messaging. Journal of Assisted Reproduction and Genetics, 39(5), 1011-1025. https://doi.org/10.1007/s10815-022-02438-8
Medeiros, C. O. (1999). Bovine sperm interaction with oviductal epithelial cells. The University of Wisconsin-Madison.
Mekata, H., & Yamamoto, M. (2022). Single-nucleotide polymor-phism on spermatogenesis associated 16 gene-coding region affecting bovine leukemia virus proviral load. Veterinary Sciences, 9(6), 275. https://doi.org/10.3390/vetsci9060275
Meyerholz, D. K., Burrough, E. R., Kirchhof, N., Anderson, D. J., & Helke, K. L. (2024). Swine models in translational research and medicine. Veterinary Pathology, 61(4), 512-523. https://doi.org/10.1177/03009858231222235
Miller, W. L. (2017). Disorders in the initial steps of steroid hormone synthesis. The Journal of Steroid Biochemistry and Molecular Biology, 165, 18-37. https://doi.org/10.1016/j.jsbmb.2016.03.009
Miyata, H., Shimada, K., Kaneda, Y., & Ikawa, M. (2024). Development of functional spermatozoa in mammalian spermiogenesis. Development, 151(14). https://doi.org/10.1242/dev.202838
Mukherjee, A., Das, P. K., Banerjee, D., & Mukherjee, J. (2023). Assisted reproductive technologies in farm animals. In P. K. Das, V. Sejian, J. Mukherjee, and D. Banerjee (Eds.), Textbook of Veterinary Physiology (pp. 615-636). Springer Nature Singapore. https://doi.org/10.1007/978-981-19-9410-4_24
Naamneh Elzenaty, R., du Toit, T., & Flück, C. E. (2022). Basics of androgen synthesis and action. Best Practice & Research Clinical Endocrinology & Metabolism, 36(4), 101665. https://doi.org/10.1016/j.beem.2022.101665
Nesbitt, C., Galina Pantoja, L., Beaton, B., Chen, C.-Y., Culbertson, M., Harms, P., Holl, J., Sosnicki, A., Reddy, S., Rotolo, M., & Rice, E. (2024b). Pigs lacking the SRCR5 domain of CD163 protein demonstrate heritable resistance to the PRRS virus and no changes in animal performance from birth to maturity. Frontiers in Genome Editing, 6, 1322012. https://doi.org/10.3389/fgeed.2024.1322012
Nikiforov, D., Grøndahl, M. L., Hreinsson, J., & Andersen, C. Y. (2022). Human oocyte morphology and outcomes of infertility treatment: a systematic review. Reproductive Sciences, 29(10), 2768-2785. https://doi.org/10.1007/s43032-021-00723-y
Owumi, S. E., Arunsi, U. O., Otunla, M. T., & Oluwasuji, I. O. (2022). Exposure to lead and dietary furan intake aggravates hypothalamus-pituitary-testicular axis toxicity in chronic experimental rats. Journal of Biomedical Research, 37(2), 100. https://doi.org/10.7555/JBR.36.20220108
Page, L., Younge, N., & Freemark, M. (2023). Hormonal determinants of growth and weight gain in the human fetus and preterm infant. Nutrients, 15(18), 4041. https://doi.org/10.3390/nu15184041
Pak, T. R., & Chung, W. C. J. (2024). Chapter 2 - Neuroendocrine control of gonadotropins in mammals. In D. O. Norris and K. H. Lopez (Eds.), Hormones and Reproduction of Vertebrates (Second Edition) (Vol. 5, pp. 23-42). Academic Press. https://doi.org/10.1016/B978-0-443-15986-2.00013-7
Park, Y., Park, Y.-B., Lim, S.-W., Lim, B., & Kim, J.-M. (2022). Time series ovarian transcriptome analyses of the porcine estrous cycle reveals gene expression changes during steroid metabolism and corpus luteum development. Animals, 12(3), 376. https://doi.org/10.3390/ani12030376
Payne, A. H., & Hales, D. B. (2004). Overview of steroidogenic enzymes in the pathway from cholesterol to active steroid hormones. Endocrine Reviews, 25(6), 947-970. https://doi.org/10.1210/er.2003-0030
Peltoniemi, O., Tanskanen, T., & Kareskoski, M. (2023). One Health challenges for pig reproduction. Molecular Reproduction and Development, 90(7), 420-435. https://doi.org/10.1002/mrd.23666
Pinto Jimenez, C. E., Keestra, S., Tandon, P., Cumming, O., Pickering, A. J., Moodley, A., & Chandler, C. I. R. (2023). Biosecurity and water, sanitation, and hygiene (WASH) interventions in animal agricultural settings for reducing infection burden, antibiotic use, and antibiotic resistance: a One Health systematic review. The Lancet Planetary Health, 7(5), e418-e434. https://doi.org/10.1016/S2542-5196(23)00049-9
Pintus, E., & Ros-Santaella, J. L. (2021). Impact of oxidative stress on male reproduction in domestic and wild animals. Antioxidants, 10(7), 1154. https://doi.org/10.3390/antiox10071154
Popli, P., Shukla, V., Kaushal, J. B., Kumar, R., Gupta, K., & Dwivedi, A. (2022). Peroxiredoxin 6 plays essential role in mediating fertilization and early embryonic development in rabbit oviduct. Reproductive Sciences, 29(5), 1560-1576. https://doi.org/10.1007/s43032-021-00689-x
Prunier, A., & Quesnel, H. (2000). Influence of the nutritional status on ovarian development in female pigs. Animal Reproduction Science, 60-61, 185-197. https://doi.org/10.1016/S0378-4320(00)00093-2
Rodriguez-Martinez, H., Roca, J., Alvarez-Rodriguez, M., & Martinez-Serrano, C. A. (2022). How does the boar epididymis regulate the emission of fertile spermatozoa? Animal Reproduction Science, 246, 106829. https://doi.org/10.1016/j.anireprosci.2021.106829
Romo-Valdez, J., Silva-Hidalgo, G., Güémez-Gaxiola, H., Romo-Valdez, A., & Romo-Rubio, J. (2022). Heat stress: influence on the physiology, productive and reproductive performance of the pig. Abanico Veterinario, 12, 1-19. https://doi.org/10.21929/abavet2022.37
Romoser, M. R., Bidne, K. L., Baumgard, L. H., Keating, A. F., & Ross, J. W. (2022). Effects of increased ambient temperature and supplemental altrenogest before pregnancy establishment in gilts. Journal of Animal Science, 100(2), skac007. https://doi.org/10.1093/jas/skac007
Rosner, M., Horer, S., Feichtinger, M., & Hengstschläger, M. (2023). Multipotent fetal stem cells in reproductive biology research. Stem Cell Research & Therapy, 14(1), 157. https://doi.org/10.1186/s13287-023-03379-4
Ruan, K.-H., & Dogne, J.-M. (2006). Implications of the molecular basis of prostacyclin biosynthesis and signaling in pharmaceutical designs. Current Pharmaceutical Design, 12(8), 925-941. https://doi.org/10.2174/138161206776055994
Samura, O., Nakaoka, Y., & Miharu, N. (2023). Sperm and oocyte chromosomal abnormalities. Biomolecules, 13(6), 1010. https://doi.org/10.3390/biom13061010
Sarandol, E., Tas, S., Serdar, Z., & Dirican, M. (2020). Effects of thiamine treatment on oxidative stress in experimental diabetes. Bratislavske Lekarske Listy, 121(3), 235-241. https://doi.org/10.4149/BLL_2020_036
Sato, R., Satoh, H., Okada, S., Goda, Y., Sueun, K., Kuroda, K., Inoue, Y., Kitahara, G., & Osawa, T. (2022). Intra-abdominal mass containing controlled internal drug release (CIDR) devices detected by CT in a Japanese Black cow. Journal of Veterinary Medical Science, 84(8), 1088-1092. https://doi.org/10.1292/jvms.22-0180
Seneda, M. M., Zangirolamo, A. F., González, S. M., & Morotti, F. (2023). Oogenesis and folliculogenesis. In Sustainable Agriculture Reviews 59: Animal Biotechnology for Livestock Production 3 (pp. 59-88). Springer. https://doi.org/10.21451/1809-3000.Rbra2021.042
Serviento, A. M., Lebret, B., & Renaudeau, D. (2020). Chronic prenatal heat stress alters growth, carcass composition, and physiological response of growing pigs subjected to postnatal heat stress. Journal of Animal Science, 98(5). https://doi.org/10.1093/jas/skaa161
Sharif, M., Hickl, V., Juarez, G., Di, X., Kerns, K., Sutovsky, P., Bovin, N., & Miller, D. J. (2022). Hyperactivation is sufficient to release porcine sperm from immobilized oviduct glycans. Scientific Reports, 12(1), 6446. https://doi.org/10.1038/s41598-022-10390-x
Shimazaki, M., Wittayarat, M., Sambuu, R., Sugita, A., Kawaguchi, M., Hirata, M., Tanihara, F., Takagi, M., Taniguchi, M., Otoi, T., & Sato, Y. (2022). Disruption of cell proliferation and apoptosis balance in the testes of crossbred cattle-yaks affects spermatogenic cell fate and sterility. Reproduction in Domestic Animals, 57(9), 999-1006. https://doi.org/10.1111/rda.14166
Sielhorst, J., Roggel-Buecker, U., Neudeck, K.-C., Kahler, A., Rohn, K., Luettgenau, J., Bollwein, H., Hollinshead, F., & Sieme, H. (2022). Effect of acetylsalicylic acid on uterine blood flow, gestation length, foal birth weight and placental weight in pregnant thoroughbred mares – a clinical pilot study. Journal of Equine Veterinary Science, 118, 104107. https://doi.org/10.1016/j.jevs.2022.104107
Smith, W. L., Urade, Y., & Jakobsson, P.-J. (2011). Enzymes of the cyclooxygenase pathways of prostanoid biosynthesis. Chemical Reviews, 111(10), 5821-5865. https://doi.org/10.1021/cr2002992
Smołucha, G., Steg, A., & Oczkowicz, M. (2024). The role of vitamins in mitigating the effects of various stress factors in pigs breeding. Animals, 14(8), 1218. https://doi.org/10.3390/ani14081218
Soede, N. M., Langendijk, P., & Kemp, B. (2011). Reproductive cycles in pigs. Animal Reproduction Science, 124(3), 251-258. https://doi.org/10.1016/j.anireprosci.2011.02.025
Spruijt, A., van Stee, L., Wolthers, K., & de Gier, J. (2022). Case report: medical management of prolonged gestation of a mummified fetus in a bitch. Frontiers in Veterinary Science, 9. https://doi.org/10.3389/fvets.2022.888807
Squires, E. J., Bone, C., & Cameron, J. (2020). Pork production with entire males: directions for control of boar taint. Animals, 10(9), 1665. https://doi.org/10.3390/ani10091665
Strauss, J. F., & FitzGerald, G. A. (2019). Chapter 4 - Steroid hormones and other lipid molecules involved in human reproduction. In J. F. Strauss and R. L. Barbieri (Eds.), Yen and Jaffe's Reproductive Endocrinology (pp. 75-114.e117). Elsevier. https://doi.org/10.1016/B978-0-323-47912-7.00004-4
Strauss, J. F., Modi, B., & McAllister, J. M. (2014). Chapter 18 - Defects in ovarian steroid hormone biosynthesis. In A. Ulloa-Aguirre and P. M. Conn (Eds.), Cellular Endocrinology in Health and Disease (pp. 285-309). Academic Press. https://doi.org/10.1016/B978-0-12-408134-5.00018-4
Sun, Y., Li, X., Jiang, C., Huang, G., Wang, J., Tian, Y., Jiang, L., Shi, X., Zhao, J., & Huang, J. (2026). Eicosapentaenoic acid improves porcine oocyte cytoplasmic maturation and developmental competence via antioxidant and mitochondrial regulatory mechanisms. Antioxidants, 15(1), 137. https://doi.org/10.3390/antiox15010137
Sutovsky, P., Hamilton, L. E., Zigo, M., Ortiz D’Avila Assumpção, M. E., Jones, A., Tirpak, F., Agca, Y., Kerns, K., & Sutovsky, M. (2024). Biomarker-based human and animal sperm phenotyping: the good, the bad and the ugly. Biology of Reproduction, 110(6), 1135-1156. https://doi.org/10.1093/biolre/ioae061
Takei, G. L. (2022). Regulation of sperm hyperactivation by transporters involved in Na+ homeostasis. Folia Pharmacologica Japonica, 157(3), 176-180. https://doi.org/10.1254/fpj.21110
Tran, Q.-K. (2020). Reciprocality between estrogen biology and calcium signaling in the cardiovascular system. Frontiers in Endocrinology, 11. https://doi.org/10.3389/fendo.2020.568203
Tucker, B. S., Craig, J. R., Morrison, R. S., Smits, R. J., & Kirkwood, R. N. (2021). Piglet viability: A review of identification and pre-weaning management strategies. Animals, 11(10), 2902. https://doi.org/10.3390/ani11102902
Tzanidakis, C., Simitzis, P., & Panagakis, P. (2023a). Precision livestock farming (PLF) systems: improving sustainability and efficiency of animal production. In F. P. García Márquez and B. Lev (Eds.), Sustainability: Cases and Studies in Using Operations Research and Management Science Methods (Vol. 333, pp. 285-337). Springer International Publishing. https://doi.org/10.1007/978-3-031-16620-4_15
Tzanidakis, C., Tzamaloukas, O., Simitzis, P., & Panagakis, P. (2023b). Precision livestock farming applications (PLF) for grazing animals. Agriculture, 13(2), 288. https://doi.org/10.3390/agriculture13020288
van den Bosch, M., Soede, N., Kemp, B., & van den Brand, H. (2023). Sow nutrition, uterine contractions, and placental blood flow during the peri-partum period and short-term effects on offspring: a review. Animals, 13(5), 910. https://doi.org/10.3390/ani13050910
van den Brand, H., Dieleman, S. J., Soede, N. M., & Kemp, B. (2000). Dietary energy source at two feeding levels during lactation of primiparous sows: I. Effects on glucose, insulin, and luteinizing hormone and on follicle development, weaning-to-estrus interval, and ovulation rate1. Journal of Animal Science, 78(2), 396-404. https://doi.org/10.2527/2000.782396x
Waberski, D., Riesenbeck, A., Schulze, M., Weitze, K. F., & Johnson, L. (2019). Application of preserved boar semen for artificial insemination: Past, present and future challenges. Theriogenology, 137, 2-7.
Wassarman, P., and Litscher, E. (2022a). Mouse zona pellucida proteins as receptors for binding of sperm to eggs. Trends in Developmental Biology, 15, 1-13.
Wassarman, P. M., & Litscher, E. S. (2022b). Female fertility and the zona pellucida. eLife, 11, e76106. https://doi.org/10.7554/eLife.76106
Wei, J., Liang, Y., Jiang, N., & Hu, G. (2022). IFN-γ differential expression in the hypothalamus-pituitary-ovary axis of thyroidectomized rats. BMC Endocrine Disorders, 22(1), 317. https://doi.org/10.1186/s12902-022-01223-z
Williams, G. L., & Cardoso, R. C. (2021). Chapter 24 - Neuroendocrine control of estrus and ovulation. In R. M. Hopper (Ed.), Bovine Reproduction (pp. 269-291). https://doi.org/10.1002/9781119602484.ch24
Yang, W.-R., Li, B.-B., Hu, Y., Zhang, L., & Wang, X.-Z. (2020). Oxidative stress mediates heat-induced changes of tight junction proteins in porcine sertoli cells via inhibiting CaMKKβ-AMPK pathway. Theriogenology, 142, 104-113. https://doi.org/10.1016/j.theriogenology.2019.09.031
Yang, Y., Jin, H., Qiu, Y., Liu, Y., Wen, L., Fu, Y., Qi, H., Baker, P. N., & Tong, C. (2022). Reactive oxygen species are essential for placental angiogenesis during early gestation. Oxidative Medicine and Cellular Longevity, 2022(1), 4290922. https://doi.org/10.1155/2022/4290922
Yu, W., Cheng, Q., & Biology, A. (2024). Accelerating yam breeding cycles with genomic selection. Genomics and Applied Biology, 15(6), 319-331.
Zaidi, S. K., Shen, W.-J., & Azhar, S. (2012). Impact of aging on steroid hormone biosynthesis and secretion. Open Longevity Science, 6, 1-30. https://doi.org/10.2174/1876326X01206010001
Zak, L. J., Gaustad, A. H., Bolarin, A., Broekhuijse, M. L. W. J., Walling, G. A., & Knol, E. F. (2017). Genetic control of complex traits, with a focus on reproduction in pigs. Molecular Reproduction and Development, 84(9), 1004-1011. https://doi.org/10.1002/mrd.22875
Zarbakhsh, S. (2021). Effect of antioxidants on preimplantation embryo development in vitro: a review. Zygote, 29(3), 179-193. https://doi.org/10.1017/S0967199420000660
Zelenkova, N., Kraus, V., Maresova, A., Pilsova, Z., Pilsova, A., Klusackova, B., Chmelikova, E., Komrskova, K., & Postlerova, P. (2025). Functional Methods for Studying Sperm-Zona Pellucida Interaction in Mammals. Methods and protocols, 8(4), 95. https://doi.org/10.3390/mps8040095
Zhang, Y.-P., Tian, L., Xie, X.-Q., Wang, Y.-T., Lyu, P., & Xi, Z.-G. (2022a). Effects of nanopolystyrene nanoplastic exposure on the development and neurotoxicity of fetal rats during gestation. Chinese Journal of Applied Physiology, 38(6), 760-765. https://doi.org/10.12047/j.cjap.6379.2022.138
Zhang, Y., Otomaru, K., Oshima, K., Goto, Y., Oshima, I., Muroya, S., Sano, M., Roh, S., & Gotoh, T. (2022b). Maternalnutrition during gestation alters histochemical properties, and mrna and microrna expression in adipose tissue of Wagyu fetuses. Frontiers in Endocrinology, 12, 797680. https://doi.org/10.3389/fendo.2021.797680
Zhang, S., Wang, D., Qi, J., Li, J., Liu, S., Sun, H., Liang, S., & Sun, B. (2026). Multiomics analysis reveals that chlorogenic acid alleviates heat stress-induced oxidative damage in prepubertal boar testes via the BLVRA-GPX3 pathway: in vivo and in vitro evidence. Journal of Animal Science and Biotechnology, 17(1), 7. https://doi.org/10.1186/s40104-025-01336-0
Zhang, L., Jiang, C., Wang, X., et al. (2024). Freezing Hu ram semen: influence of different penetrating cryoprotectants and egg yolk level on the post-thaw quality of sperm. Anim Biosci, 37(9), 1548-1557. https://doi.org/10.5713/ab.24.0167
Zhao, H., Wang, Y., & Yang, Y. (2023a). Follicular development and ovary aging: single-cell studies. Biology of Reproduction, 109(4), 390-407. https://doi.org/10.1093/biolre/ioad080
Zhao, W., Lai, X., Liu, D., Zhang, Z., Ma, P., Wang, Q., Zhang, Z., & Pan, Y. (2020). Applications of support vector machine in genomic prediction in pig and maize populations. Frontiers in Genetics, 11. https://doi.org/10.3389/fgene.2020.598318
Zhao, W., Zhang, Z., Ma, P., Wang, Z., Wang, Q., Zhang, Z., & Pan, Y. (2023b). The effect of high-density genotypic data and different methods on joint genomic prediction: A case study in large white pigs. Animal Genetics, 54(1), 45-54. https://doi.org/10.1111/age.13275
Zheng, Y., Gao, Q., Li, T., Liu, R., Cheng, Z., Guo, M., Xiao, J., Wu, D., & Zeng, W. (2022). Sertoli cell and spermatogonial development in pigs. Journal of Animal Science and Biotechnology, 13(1), 45. https://doi.org/10.1186/s40104-022-00687-2
Zhi, M., Zhang, J., Tang, Q., Yu, D., Gao, S., et al. (2022). Generation and characterization of stable pig pregastrulation epiblast stem cell lines. Cell Research, 32(4), 383-400. https://doi.org/10.1038/s41422-021-00592-9
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Scientia Agropecuaria

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
The authors who publish in this journal accept the following conditions:
a. The authors retain the copyright and assign to the magazine the right of the first publication, with the work registered with the Creative Commons attribution license, which allows third parties to use the published information whenever they mention the authorship of the work and the First publication in this journal.
b. Authors may make other independent and additional contractual arrangements for non-exclusive distribution of the version of the article published in this journal (eg, include it in an institutional repository or publish it in a book) as long as it clearly indicates that the work Was first published in this journal.
c. Authors are encouraged to publish their work on the Internet (for example, on institutional or personal pages) before and during the review and publication process, as it can lead to productive exchanges and a greater and faster dissemination of work Published (see The Effect of Open Access).




