Conversion of coffee and plastics residues into activated carbon by chemical activation with ammonia
DOI:
https://doi.org/10.17268/sci.agropecu.2026.029Palabras clave:
Activated carbon, pyrolysis, biomass residues, plastic wasteResumen
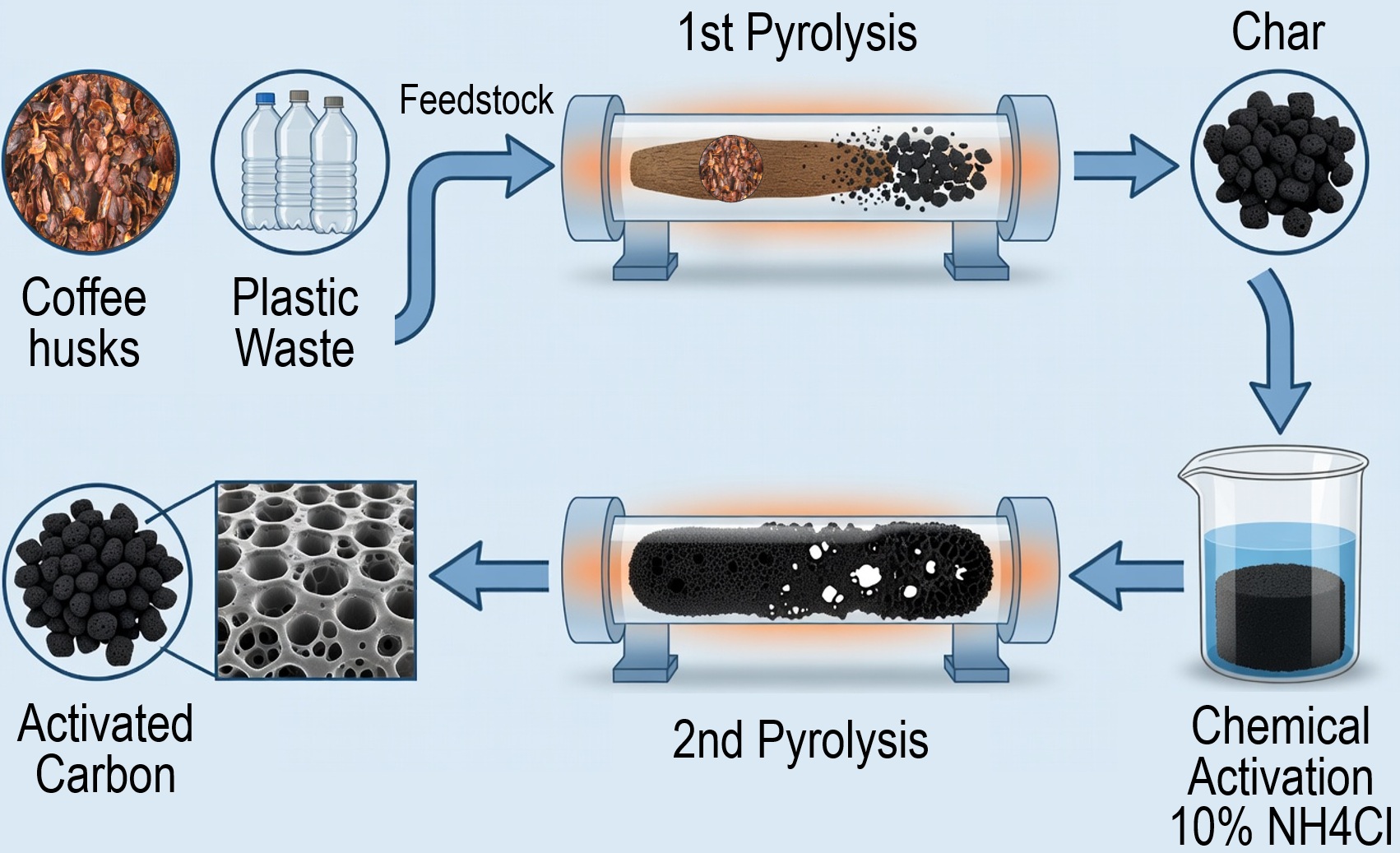
Aiming for the valorization of agricultural biomass residues such as coffee husks and plastics wastes such as polyethylene terephthalate (PET), a method for transforming them into activated carbon by using ammonia chloride as activation agent is presented in this study. Firstly, mixtures of PET waste were processed through a pyrolytic process at different operational conditions through a factorial experimental design. The obtained material with the best characteristics of specific surface area (SSA) and mass yield was obtained at 600 °C and 2.5 hours of treatment. These parameters were then used for performing the activation of coffee husks residues and mixtures of them with PET (in mass rates of 1:1 and 1:2 of PET and biomass, respectively) All the obtained materials were then characterized through a Scanning Electron Microscopy - Energy Dispersive Spectroscopy (SEM–EDS), X-Ray Fluorescence (XRF), X-Ray Diffraction (XRD) and a Differential Scanning Calorimetry - Thermogravimetric Analysis (DSC–TGA) technique, and other physicochemical properties (particle size, bulk density, humidity content and ash content) were also measured, all properties were then compared with characteristics of commercial activated carbon. The activated carbon obtained from coffee husks residues showed a low specific surface area (SSA) and a higher pyrolysis yield. The mixtures of biomass and PET showed lower SSA and higher pyrolysis yield than the ones obtained from PET. The mixture of 1:1 is the best on SSA and yield. The textural analysis was performed (BET, BJH, XRF, XRD and SEM–EDS) on the samples of pure pet and mixture 1:1. This shows that the method allows the utilization of coffee and PET residues to produce activated carbon in a more sustainable way respect to other methods of activation (the proposed method uses low mass relations 10% and no postprocessing).
Citas
Block, C., Ephraim, A., Weiss-Hortala, E., Minh, D. P., Nzihou, A., & Vandecasteele, C. (2019). Co-pyrogasification of Plastics and Biomass, a Review. Waste and Biomass Valorization, 10(3), 483–509. https://doi.org/10.1007/s12649-018-0219-8
Cai, X., Ren, W., Li, C., Li, Q., & Hu, X. (2026). Synergistic CO2-K2C2O4 activation of furfural residue toward high-adsorption activated carbon for phenol removal. Journal of Analytical and Applied Pyrolysis, 193, 107408. https://doi.org/10.1016/j.jaap.2025.107408
Chia, J. W. F., Sawai, O., & Nunoura, T. (2020). Reaction pathway of poly(ethylene) terephthalate carbonization: Decomposition behavior based on carbonized product. Waste Management, 108, 62–69. https://doi.org/10.1016/j.wasman.2020.04.035
Ciner, M. N., Özbaş, E. E., Ozcan, H. K., & Ongen, A. (2026). Potential of Physical Activated Carbon Derived from Pyrolyzed Waste Coffee Grounds as an Adsorbent for Dye Removal. Water, Air, and Soil Pollution, 237(9). https://doi.org/10.1007/s11270-026-09235-4
González-García, P. (2018). Activated carbon from lignocellulosics precursors: A review of the synthesis methods, characterization techniques and applications. Renewable and Sustainable Energy Reviews, 82, 1393–1414. https://doi.org/10.1016/J.RSER.2017.04.117
Guimarães, T., de Carvalho Teixeira, A. P., de Oliveira, A. F., & Lopes, R. P. (2020). Biochars obtained from arabica coffee husks by a pyrolysis process: characterization and application in Fe(II) removal in aqueous systems. New Journal of Chemistry, 44(8), 3310–3322. https://doi.org/10.1039/C9NJ04144C
Illingworth, J. M., Rand, B., & Williams, P. T. (2022). Understanding the mechanism of two-step, pyrolysis-alkali chemical activation of fibrous biomass for the production of activated carbon fibre matting. Fuel Processing Technology, 235, 107348. https://doi.org/10.1016/j.fuproc.2022.107348
López Nina, L. G., Valdez, S. O., Cabrera, S. O., Martinez, G. X., Surculento, R., Lara, R. M., Velasco, J. A., Torres, T. G., Tirado, N. S., & Zambrana, S. (2021). Conversión de residuos domiciliarios de bioseguridad y residuos de poda en productos útiles: bioachar y carbón activado. Universidad Mayor de San Andrés.
Marsh, H., & Rodríguez-Reinoso, F. (2006). Activated carbon. Elsevier.
MELLIFIQ. (2025). Activated Carbon Datasheets | Mellifiq. Https://Mellifiq.Com/En/. https://mellifiq.com/en/activated-carbon-technical-datasheet/
Mochizuki, Y., Bud, J., Byambajav, E., & Tsubouchi, N. (2025). Pore properties and CO2 adsorption performance of activated carbon prepared from various carbonaceous materials. Carbon Resources Conversion, 8(1). https://doi.org/10.1016/j.crcon.2024.100237
Moussavi, G., Alahabadi, A., Yaghmaeian, K., & Eskandari, M. (2013). Preparation, characterization and adsorption potential of the NH4Cl-induced activated carbon for the removal of amoxicillin antibiotic from water. Chemical Engineering Journal, 217, 119–128. https://doi.org/10.1016/j.cej.2012.11.069
Newar, R., Sultana, N., & Baruah, A. (2026). Nanoarchitectonics of waste rice derived SiO2@Activated carbon composite for high-performance adsorptive removal of CO2 and cationic dyes. Journal of Solid State Chemistry, 354, 125733. https://doi.org/10.1016/j.jssc.2025.125733
Nguyen, M. L., Ngo, H. L., Truong, B. N., & Nguyen, T. T. (2026). Soybean-residue-derived H3PO4-activated Carbon For High-Efficiency Adsorption of Hexavalent Chromium: Optimization, Characterization, and Adsorption Mechanisms. Water, Air, & Soil Pollution, 237(3), 174. https://doi.org/10.1007/s11270-025-08828-9
Paradela, F., Pinto, F., Ramos, A. M., Gulyurtlu, I., & Cabrita, I. (2009). Study of the slow batch pyrolysis of mixtures of plastics, tyres and forestry biomass wastes. Journal of Analytical and Applied Pyrolysis, 85(1–2), 392–398. https://doi.org/10.1016/j.jaap.2008.09.003
Rajadurai, B., & Chandradass, J. (2024). Mechanical, thermal, and morphological characterization of polylactic acid composites reinforced with coconut shell activated carbon. Materials Research Express, 11(11). https://doi.org/10.1088/2053-1591/ad95e6
Ramirez, N., Sardella, F., Deiana, C., Schlosser, A., Müller, D., Kißling, P. A., Klepzig, L. F., & Bigall, N. C. (2020). Capacitive behavior of activated carbons obtained from coffee husk. RSC Advances, 10(62), 38097–38106. https://doi.org/10.1039/d0ra06206e
Riyanto, C. A., Ampri, M. S., & Martono, Y. (2020). Synthesis and characterization of nano activated carbon from annatto peels (Bixa orellana L.) viewed from temperature activation and impregnation ratio of H3PO4. EKSAKTA: Journal of Sciences and Data Analysis, 1(1), 44–50.
Şahin, Ö., Saka, C., Ceyhan, A. A., & Baytar, O. (2016). The pyrolysis process of biomass by two-stage chemical activation with different methodology and iodine adsorption. Energy Sources, Part A: Recovery, Utilization, and Environmental Effects, 38(12), 1756–1762. https://doi.org/10.1080/15567036.2014.956195
Scheirs, J., & Kaminsky, W. (2006). Feedstock recycling and pyrolysis of waste plastics. In Focus on Catalysts (Vol. 2006, Number 9). https://doi.org/10.1016/s1351-4180(06)71853-0
Serafin, J. (2017). Utilization of spent dregs for the production of activated carbon for CO2 adsorption. Polish Journal of Chemical Technology, 19(2), 44–50. https://doi.org/10.1515/pjct-2017-0026
Serafin, J., Antosik, A. K., Wilpiszewska, K., & Czech, Z. (2018). Preparation of Activated Carbon from the Biodegradable film for Co2 Capture Applications. Polish Journal of Chemical Technology, 20(3), 75–80. https://doi.org/10.2478/pjct-2018-0041
Serafin, J., & Dziejarski, B. (2023). Application of isotherms models and error functions in activated carbon CO2 sorption processes. Microporous and Mesoporous Materials, 354, 112513. https://doi.org/10.1016/j.micromeso.2023.112513
Shafeeyan, M. S., Daud, W. M. A. W., Shamiri, A., & Aghamohammadi, N. (2015). Modeling of Carbon Dioxide Adsorption onto Ammonia-Modified Activated Carbon: Kinetic Analysis and Breakthrough Behavior. Energy & Fuels, 29(10), 6565–6577. https://doi.org/10.1021/acs.energyfuels.5b00653
Surculento Villalobos, R., Marín M., G. M., & Lopez N, L. (2023). Obtención de carbón activado a partir de mezclas de residuos de plásticos pet por activación con ácido fosfórico. Revista Boliviana de Química, 40(3). https://doi.org/10.34098/2078-3949.40.3.3
Descargas
Publicado
Cómo citar
Número
Sección
Licencia
Derechos de autor 2026 Scientia Agropecuaria

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial 4.0.
Los autores que publican en esta revista aceptan los siguientes términos:
a. Los autores conservan los derechos de autor y conceden a la revista el derecho publicación, simultáneamente licenciada bajo una licencia de Creative Commons que permite a otros compartir el trabajo, pero citando la publicación inicial en esta revista.
b. Los autores pueden celebrar acuerdos contractuales adicionales separados para la distribución no exclusiva de la versión publicada de la obra de la revista (por ejemplo, publicarla en un repositorio institucional o publicarla en un libro), pero citando la publicación inicial en esta revista.
c. Se permite y anima a los autores a publicar su trabajo en línea (por ejemplo, en repositorios institucionales o en su sitio web) antes y durante el proceso de presentación, ya que puede conducir a intercambios productivos, así como una mayor citación del trabajo publicado (ver efecto del acceso abierto).




