Antagonistic and growth-enhancing effect of native Trichoderma spp. on ‘Gros Michel’ banana under Foc R1 infection
Efecto antagónico y promotor del crecimiento de especies nativas de Trichoderma en el banano ‘Gros Michel’ bajo infección por Foc R1
Pedro Terrero Yepez1 *; Rafaela Mayorga Morejon1; Paola Rodulfo Acuña1; Karina Solis Hidalgo1; Arianna Mendoza Intriago2; Nicole Factos Laiño1
1 Estación Experimental Tropical Pichilingue, Instituto Nacional de Investigaciones Agropecuarias, Programa Nacional de Banano, Plátano y otras Musáceas, km 5 vía Quevedo-El Empalme, Cantón Mocache, Los Ríos, Ecuador.
2 Carrera de Agronomía, Facultad de Ciencias Agrarias y Forestales, Universidad Técnica Estatal de Quevedo, km 1.5 a Santo Domingo de los Tsáchilas, Quevedo, Ecuador.
ORCID de los autores:
P. Terrero Yepez: https://orcid.org/0000-0002-4492-4577 R. Mayorga Morejon: https://orcid.org/0000-0003-2002-3122
P. Rodulfo Acuña: https://orcid.org/0009-0007-4697-2752 K. Solis Hidalgo: https://orcid.org/0000-0002-2696-4540
A. Mendoza Intriago: https://orcid.org/0009-0002-7575-9727 N. Factos Laiño: https://orcid.org/0000-0002-1484-2669
ABSTRACT
Banana production in Ecuador has been severely impacted by Fusarium wilt, caused by Fusarium oxysporum f. sp. cubense (Foc). This persistent soilborne pathogen remains viable for decades and reduces the effectiveness of conventional control strategies. Trichoderma spp. has emerged as a promising biological alternative due to its antagonistic capacity and plant-associated effects. This study evaluated the efficacy of native Trichoderma strains as biological control agents against Foc Race 1 (R1) in Musa AAA cv. Gros Michel. Dual culture assays and greenhouse experiments were conducted using a completely randomized design, with data analyzed by ANOVA and Tukey’s test (α = 0.05). In vitro, T. lixii, T. asperellum, and T. harzianum inhibited up to 58% of Foc radial growth. Under greenhouse conditions, Trichoderma application significantly reduced vascular wilt severity (p < 0.05) and was associated with increased pseudostem diameter and root biomass. T. afroharzianum and T. lixii lowered disease incidence to levels comparable to those of the healthy control. These findings support the potential use of native Trichoderma isolates in sustainable management strategies for Fusarium wilt in banana.
Keywords: Biological control; fungal diseases; rhizosphere microbiota; integrated disease management; Musaceae.
RESUMEN
La producción de banano en Ecuador se ha visto gravemente afectada por la marchitez por Fusarium, causada por Fusarium oxysporum f. sp. cubense (Foc). Este patógeno persistente del suelo permanece viable durante décadas y reduce la efectividad de las estrategias de control convencionales. Trichoderma spp. ha surgido como una alternativa biológica prometedora debido a su capacidad antagónica y efectos asociados a la planta. Este estudio evaluó la eficacia de cepas nativas de Trichoderma como agentes de control biológico contra Foc Raza 1 (R1) en Musa AAA cv. Gros Michel. Se realizaron ensayos de cultivo dual y experimentos en invernadero utilizando un diseño completamente aleatorio, con datos analizados mediante ANOVA y la prueba de Tukey (α = 0,05). In vitro, T. lixii, T. asperellum y T. harzianum inhibieron hasta el 58% del crecimiento radial de Foc. En condiciones de invernadero, la aplicación de Trichoderma redujo significativamente la severidad de la marchitez vascular (p < 0,05) y se asoció con un aumento del diámetro del pseudotallo y la biomasa radicular. T. afroharzianum y T. lixii redujeron la incidencia de la enfermedad a niveles comparables a los del grupo control sano. Estos hallazgos respaldan el uso potencial de aislados nativos de Trichoderma en estrategias de manejo sostenible para la marchitez por Fusarium en banano.
Palabras clave: Control biológico; enfermedades fúngicas; microbiota de la rizosfera; manejo integrado de enfermedades; Musaceae.
1. Introduction
Fusarium oxysporum f. sp. cubense Race 1 (Foc R1) is one of the majors phytopathogens affecting banana (Musa spp., Musaceae) and remains a recurring and evolving threat to global production (Izquierdo-García et al., 2024; Kema et al., 2021). The predominant method for managing phytopathogenic fungi has traditionally been the application of synthetic fungicides (Torres-Rodríguez et al., 2024). However, their intensive use alters the physicochemical and biological properties of soils, reduces fertility, contaminates air and water, and leads to pesticide residues in food with potential risks to human health (Anis Mufida et al., 2024; Torres-Rodríguez et al., 2024; Sánchez-Alarcón et al., 2021). Regarding Foc R1, fungicide-based strategies are largely ineffective because of the pathogen’s biology and its long-term persistence in soil (Ploetz, 2015). Consequently, sustainable agriculture has emerged as a key alternative to reduce agrochemical dependence while maintaining productivity and protecting natural resources, human health, and ecosystem balance (Zou et al., 2024).
Beyond the current impact of Fusarium oxysporum f. sp. cubense Race 1, banana production in Ecuador faces an increasing phytosanitary risk associated with the potential introduction of Tropical Race 4 (Foc TR4), one of the most destructive pathogens threatening global banana systems. Ecuador, as the world’s leading banana exporter, remains particularly vulnerable due to the extensive monoculture of susceptible cultivars and the limited availability of effective curative measures once the pathogen becomes established. Although this study focuses on Foc R1, research on biological control agents and plant–microbe interactions is highly relevant to TR4 preparedness, as preventive strategies based on microbial-mediated disease suppression and enhanced plant resilience are among the few sustainable tools available for long-term management. In this sense, studies evaluating native antagonistic microorganisms contribute not only to current disease control but also to strengthening national biosecurity and risk mitigation frameworks.
Within this framework, the use of antagonistic microorganisms has gained relevance as a biocontrol strategy, given their ability to suppress pathogens and promote plant growth in economically important crops (Zheng et al., 2024). Among them, Trichoderma spp. has been widely incorporated into integrated pest and disease management programs in banana due to its ability to colonize the rhizosphere, enhance nutrient uptake, stimulate plant growth, and activate systemic defense responses (Harman et al., 2021). The mechanisms of action of Trichoderma include mycoparasitism, secretion of secondary metabolites, antibiotic production, and competition for nutrients and space (Esquivel-Naranjo et al., 2025; Natsiopoulos et al., 2024; Chávez-Avilés et al., 2024). These properties position Trichoderma as a key tool in the transition toward more sustainable production systems by reducing dependence on synthetic fungicides and mitigating their environmental impacts.
Recent studies have reported the antagonistic potential of Trichoderma spp. strains against Fusarium oxysporum, associated with enzyme production and reduced pathogen growth under in vitro conditions (Nguyen Quoc Khuong et al., 2023), as well as with antifungal traits and improvements in plant health reported in cropping systems (Jamil, 2021).
Despite the extensive use of Trichoderma spp. as biological control agents, the relationship between direct antagonism observed under in vitro conditions and effective disease suppression in planta remains unclear, particularly for Fusarium wilt of banana. Many studies rely primarily on in vitro assays, which may not adequately capture the complexity of plant-microbe-pathogen interactions occurring in the rhizosphere. Consequently, isolates exhibiting strong in vitro antagonism do not always translate into superior performance under greenhouse or field conditions.
Addressing this gap is critical for the rational selection of biological control agents. Therefore, the objective of this study was to evaluate native Trichoderma spp. isolates from Ecuador by integrating in vitro antagonism assays with greenhouse experiments of disease suppression and plant growth promotion in Musa AAA cv. Gros Michel infected with Foc R1.
2. Methodology
The plant material comprised banana plantlets (Musa AAA cv. Gros Michel, family Musaceae) produced by in vitro propagation and acclimatized for eight weeks under controlled greenhouse conditions. The plantlets were generated and supplied by the Biotechnology Department of the National Institute of Agricultural Research (INIAP), which develops certified propagation protocols for Musaceae.
The plants were grown in plastic pots containing a homogeneous substrate composed of agricultural soil and sand (2:1, v/v). The substrate was steam-sterilized prior to use to reduce background microbial interference and ensure that observed effects were primarily attributable to the inoculated Trichoderma isolates and Foc R1. Pots were maintained under greenhouse conditions and irrigated as required to avoid water stress throughout the experimental period.
The biological material consisted of seven native Trichoderma spp. isolated from the INIAP microbial collection (Table 1), previously identified at the molecular level and cryopreserved at −80 °C. The molecular identification was performed at the Biotechnology Department of the Tropical Experimental Station Pichilingue (INIAP) through ITS and TEF1-α gene sequencing and phylogenetic analysis. These identification results were previously published by our research group (Terrero-Yépez et al., 2025), where sequences showing >99% identity with GenBank reference strains confirmed their taxonomic classification.
Antagonistic activity was assessed through dual culture assays on PDA, following Silva (2018) with modifications. A 5-mm disk of actively growing Foc R1 mycelium was placed at one edge of the plate and a 5-mm disk of the Trichoderma isolate on the opposite edge. Each treatment included four replicates.
Plates were incubated at 25 ± 1 °C under low-light conditions. Radial growth was measured every 24 h. Percentage of inhibition was calculated as: PRI (%) = 100 × (R − r) / R, where R is the pathogen radius in the control and r is the radius in dual culture.
In vivo evaluation under greenhouse conditions
A total of 135 banana plantlets were assigned to nine treatments (15 plants per treatment). Trichoderma suspensions (200 mL per plant at 1 × 10⁷ conidia mL⁻¹) were applied at 15-day intervals for three applications. After 90 days sowing (DAS), plants were inoculated with Foc R1 using 5-mm mycelial plugs placed into the root zone.
Evaluated variables: Plant height (cm), Pseudo-stem diameter (cm) measured 2 cm above the base using a digital caliper, Number of functional leaves, and Root fresh and dry biomass (g) (drying at 70 °C for 72 h).
Disease incidence and severity
Severity was assessed monthly using a 6-level external symptom scale (Orjeda, 1998):
1 = no symptoms;
2 = yellowing of older leaves;
3 = yellowing of lower leaves;
4 = yellowing of young leaves;
5 = severe yellowing;
6 = dead plant.
Experimental design and statistical analysis
A completely randomized design (CRD) was used for both in vitro and in vivo experiments. ANOVA was performed after verifying normality and homoscedasticity. Means were compared using Tukey’s test at α = 0.05.
Statistical analyses were conducted using R software (version 4.3.1) (packages stats and agricolae).
3. Results and discussion
In vitro antagonism of Trichoderma spp. against Foc R1
Dual culture assays revealed marked differences among the seven Trichoderma isolates, demonstrating that antagonism is a strain-dependent trait. After 48 h of incubation, T. lixii (F17) exhibited the strongest suppressive activity, inhibiting the radial growth of Foc R1 by 58%. This substantial reduction indicates an efficient combination of competition for space and nutrients, as well as the possible production of antifungal metabolites. T. asperellum (F74) and T. afroharzianum (F19) followed with inhibition values of 45% and 38%, respectively, constituting the group of intermediate antagonists.
Table 1
Trichoderma spp. isolates from INIAP collection
| Trichoderma species | Geographic origin | Botanical source | Physical source |
F74 | T. asperellum | Guayas | Plantain | Soil |
F73 | T. lentiforme | Santa Elena | Banana | Root |
F84 | T. harzianum | Los Ríos | Banana | Soil |
F78 | T. afroharzianum | Los Ríos | Banana | Soil |
F17 | T. lixii | Manabí | Plantain | Leaf |
F19 | T. afroharzianum | Santo Domingo | Abacá | Soil |
F76 | T. asperellum | Los Ríos | Banana | Soil |
Moderate inhibition levels were observed for T. harzianum (F84; 34%) and another T. asperellum isolate (F76; 31%). In contrast, both isolates of T. afroharzianum (F78 and F19) displayed inhibition values below 25%, indicating a limited suppressive effect under direct confrontation. This weaker in vitro antagonism is consistent with previously reported patterns for some endophytic Trichoderma strains, which may rely more on indirect mechanisms, such as the induction of plant defenses, than on direct competition.
The dual-culture assays confirmed that the antagonistic activity of Trichoderma against Fusarium oxysporum f. sp. cubense R1 is strongly strain-dependent, even among isolates within the same species (Rodríguez-García et al., 2020; Kumari et al., 2024). While isolates such as T. lixii (F17) exhibited strong direct inhibition, others showed moderate or low suppressive effects under in vitro conditions. Importantly, reduced inhibition in dual culture assays should not be interpreted as low biocontrol potential, as increasing evidence indicates that several Trichoderma strains rely primarily on indirect mechanisms, including induction of systemic resistance, modulation of plant defense pathways, and efficient root colonization, which are not captured in host-free assays (Chen et al., 2021; Natsiopoulos et al., 2024). Therefore, in vitro confrontation tests provide only partial insight into the biocontrol capacity of Trichoderma, reinforcing the need to integrate laboratory assays within planta evaluations to achieve a biologically meaningful selection of candidate strains (Fotoohiyan et al., 2017; Chávez-Avilés et al., 2024).
Suppression of disease severity, incidence, and infection intensity in biotized banana plants
In the treatment inoculated with Foc R1, plants developed pronounced disease symptoms, reaching a mean severity score of 4.67 on the six-point scale. In contrast, plants treated with Trichoderma showed much lower severity values, ranging from 1.47 to 2.53 (p < 0.0001). These reductions indicate that the isolates slowed disease progression and limited typical wilt symptoms, including leaf yellowing and vascular decline.
Disease incidence followed the same trend. Foc R1 infected 100% of the plants in positive control, confirming the pathogen's high aggressiveness. In contrast, incidence in the Trichoderma-treated plants was markedly lower, ranging from 6.67% to 66.67%, depending on the isolate. This reduction demonstrates that the antagonistic strains were able to limit infection and prevent the spread of vascular wilt among the plants.
Infection intensity and mean grade further supported these findings. The pathogen control recorded 92% infection intensity and a mean grade of 4.60, reflecting both extensive tissue colonization and severe symptom progression. Trichoderma treatments significantly reduced both indicators by 24% – 50%, and F78 achieved a mean grade of only 1.22, confining symptoms to the mildest categories. These results confirm that Trichoderma not only limits the number of infected plants but also restricts disease progression within individual plants. As shown in Table 2, all Trichoderma isolates significantly reduced disease severity and infection parameters compared with the pathogen control.
Under the experimental conditions evaluated, native Trichoderma spp. proved to be a promising biological alternative for the management of Fusarium wilt in ‘Gros Michel’ banana, as evidenced by the significant reductions in disease severity, incidence, and infection intensity compared with the pathogen control. These results support the initial hypothesis that biotization with Trichoderma mitigates disease expression and improves plant performance, while also revealing marked strain-specific differences.
Table 2
Disease severity and infection index in plants treated with native Trichoderma spp. Isolates
Treatment | Severity (Media ± E.E.) | Tukey Severity | % Infección (Mean± S.E.) | Tukey Infection index |
Foc | 4.67 ± 0.25 | A | 100.00 ± 6.89 | A |
F74 | 2.53 ± 0.25 | B | 66.67 ± 6.89 | A B |
F19 | 2.40 ± 0.25 | B | 66.67 ± 6.89 | A B |
F17 | 2.20 ± 0.25 | B | 53.33 ± 6.89 | B |
F73 | 1.73 ± 0.25 | B C | 40.00 ± 6.89 | B C |
F84 | 1.73 ± 0.25 | B C | 46.67 ± 6.89 | B |
F76 | 1.53 ± 0.25 | B C | 53.33 ± 6.89 | B |
F78 | 1.47 ± 0.25 | B C | 6.67 ± 6.89 | C D |
Water | 1.00 ± 0.25 | C | 0.00 ± 6.89 | D |
Means followed by the same letter within a column are not significantly different according to Tukey’s test (p ≤ 0.05).
Such variability is consistent with the broad functional diversity described within the genus Trichoderma, which includes mechanisms such as competition, mycoparasitism, antibiosis, and induction of host defenses (Harman et al., 2021; Esquivel-Naranjo et al., 2025). The strong suppression of disease parameters observed across isolates indicates that, under greenhouse conditions, Trichoderma can effectively limit both pathogen establishment and symptom progre-ssion, reinforcing its role as a disease-modulating agent rather than a strictly antagonistic one.
Integration of in vitro and in vivo results further highlighted that direct antagonism does not necessarily predict greenhouse performance. While F17 showed the strongest inhibition of Foc R1 in dual-culture assays, F78 emerged as the most effective isolate in planta, achieving near-complete suppression of disease incidence and markedly reducing severity and infection intensity to levels comparable to those of the healthy control. This pattern aligns with previous studies showing that isolates selected for in vitro antagonism often exhibit disproportionate or contrasting performance under greenhouse conditions, where indirect mechanisms and plant-mediated interactions play a dominant role (Fotoohiyan et al., 2023; Natsiopoulos et al., 2024; Chávez-Avilés et al., 2024). The superior performance of F78 suggests that mechanisms such as efficient root colonization, induction of systemic resistance, or enhanced plant tolerance to vascular dysfunction may be more relevant than direct mycoparasitism in suppressing Fusarium wilt. These findings emphasize the necessity of combining laboratory and greenhouse evaluations to identify robust biocontrol candidates and underscore the value of native isolates, which may be better adapted to local agroecological conditions and therefore more reliable for integration into sustainable disease management programs.
Plant growth responses under biotic stress from Foc R1
Growth parameters differed significantly among treatments (Table 3). Foc R1 infection substantially compromised plant vigor, as shown by the lowest pseudostem diameter (1.29 cm) and reduced biomass in the pathogen control. In contrast, all Trichoderma treatments maintain pseudostem diameters above 2.27 cm, values comparable to those of the healthy control. This parameter, closely associated with structural stability and resource allocation in Musaceae, indicates that Trichoderma mitigated vascular obstruction and promoted tissue integrity despite pathogen pressure.
Shoot biomass exhibited pronounced differences among treatments. Plants inoculated only with Foc R1 accumulated the lowest biomass (76.33 g), whereas those treated with the strains F78 and F17 reached 151.67 g and 151.33 g, respectively, approximately double that of the pathogen control. The statistical overlap among most Trichoderma treatments highlights the genus's generally strong biostimulatory capacity, often associated with enhanced nutrient uptake, improved root functionality, and phytohormone-like activity.
Root fresh weight followed the same trend. F78 produced the highest biomass (96.67 g), followed by F17 and F73, whereas the pathogen control had the lowest value (57 g). These results indicate that root colonization by Trichoderma promotes root proliferation even under pathogen stress, thereby improving water and nutrient acquisition and contributing to plant resilience.
Native Trichoderma isolates mitigated the detrimental effects of Fusarium wilt on banana plant development. Infection with Foc R1 markedly reduced pseudostem diameter and biomass accumulation, consistent with the vascular obstruction and impaired resource translocation characteristic of Fusarium wilt in banana (Ploetz, 2015; Kema et al., 2021).
Table 3
Integrated effects of native Trichoderma spp. on growth-related variables of ‘Gros Michel’ banana under Foc R1 infection
Treatment | Pseudostem diameter (cm) | Tukey | Shoot fresh weight (g) | Tukey | Root fresh weight (g) | Tukey | Plant height (cm) | Tukey |
Water | 2.38 | A | 127.33 | B | 70.33 | BC | 16.96 | AB |
F74 | 2.33 | A | 144.00 | A | 85.67 | AB | 17.22 | AB |
F17 | 2.33 | A | 151.33 | A | 91.33 | AB | 16.96 | AB |
F84 | 2.31 | A | 147.33 | A | 83.33 | AB | 17.36 | AB |
F73 | 2.31 | A | 148.67 | A | 88.00 | AB | 17.64 | A |
F19 | 2.31 | A | 146.33 | A | 86.33 | AB | 16.97 | AB |
F76 | 2.31 | A | 125.67 | B | 70.67 | BC | 15.98 | BC |
F78 | 2.27 | A | 151.67 | A | 96.67 | A | 15.86 | BC |
Foc R1 | 1.29 | B | 76.33 | C | 57.00 | C | 15.16 | C |
In contrast, all Trichoderma treatments maintain pseudostem diameters comparable to the healthy control, suggesting preservation of vascular functionality and structural stability. Similar protective effects on plant architecture under Fusarium stress have been reported for Trichoderma asperellum and T. koningiopsis in banana, where treated plants showed improved stem robustness and biomass accumulation despite pathogen presence (Luo et al., 2023; Sarma et al., 2025). These findings support the notion that Trichoderma can alleviate the physiological constraints imposed by Foc The pronounced increases in shoot and root biomass, particularly in plants treated with isolates F78 and F17, further highlight the dual role of Trichoderma as a biocontrol and plant growth–promoting agent under biotic stress. Root biomass preservation is especially relevant, as early root deterioration is a key driver of Fusarium wilt progression in banana (Kema et al., 2021). Enhanced root development under pathogen pressure has been widely linked to Trichoderma-mediated improvements in nutrient mobilization, root system architecture, and modulation of plant hormonal balance (Contreras-Cornejo et al., 2024; Yao et al., 2023). Although plant height differences were less pronounced, several isolates partially offset the growth restriction caused by Foc R1, indicating that vertical development is less sensitive than biomass-related traits but still responsive to fungal biostimulation, rather than merely suppressing symptom expression.
Integration of antagonism and plant growth promotion patterns
Overall, these results demonstrate that in vitro antagonism alone is not a reliable predictor of disease suppression or plant growth promotion under greenhouse conditions, highlighting the importance of host-mediated and indirect mechanisms in the biocontrol performance of Trichoderma (Figure 1). Differences among isolates suggest that traits such as efficient root colonization, production of bioactive metabolites, and induction of systemic resistance may play a more prominent role in vivo than direct mycoparasitism observed in dual culture assays. Consequently, strain-specific performance must be evaluated through integrated laboratory and greenhouse approaches to identify robust candidates for Fusarium wilt management.
An additional contribution of this study lies in its regional relevance. Although Trichoderma spp. has been widely studied worldwide, information on the performance of Ecuadorian native isolates against Fusarium oxysporum f. sp. cubense R1 remains limited. This work addresses that gap by identifying native strains, particularly T. afroharzianum F78, that combine strong disease suppression with plant growth promotion under controlled conditions. The use of local isolates is especially valuable, as they may be better adapted to regional soils, climatic conditions, and banana genotypes, potentially enhancing their stability and effectiveness in production systems.
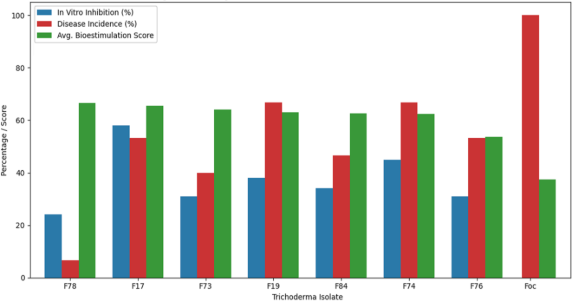
Figure 1. Integrated performance of native Trichoderma spp. isolates against Foc R1 in ‘Gros Michel’ banana. Radar plot summarizing normalized values of in vitro antagonism, disease severity reduction, disease incidence reduction, and plant growth promotion under greenhouse conditions. Higher values indicate superior overall performance.
Despite the promising results obtained, some limitations should be considered when interpreting the findings. The experiments were conducted exclusively under greenhouse conditions using a single banana cultivar (‘Gros Michel’) and a single F. oxysporum f. sp. cubense R1 isolate, which may not fully represent the diversity of host–pathogen interactions present under field conditions. In addition, the use of steam-sterilized substrates likely reduced background microbial interactions, potentially amplifying the apparent effects of Trichoderma isolates compared with non-sterile soils. Therefore, extrapolation of these results to commercial systems should be made with caution.
Future research should focus on elucidating the mechanisms underlying strain-specific behavior through analyses of root colonization dynamics, expression of defense-related genes in banana, and characterization of antifungal metabolites. Moreover, long-term field evaluations across different agroecological conditions are essential to determine the consistency and practical relevance of these effects. Such efforts will be critical for the development of sustainable, biologically based strategies for Fusarium wilt management in Ecuador and other tropical banana-producing regions.
4. Conclusions
Native Trichoderma spp. isolates significantly reduced disease severity, incidence, and infection intensity caused by Fusarium oxysporum f. sp. cubense R1 in ‘Gros Michel’ banana under greenhouse conditions.
In vitro antagonistic activity did not necessarily predict greenhouse performance, highlighting the importance of indirect mechanisms such as root colonization and induction of plant defense responses.
Selected isolates, particularly F78 and F17, not only suppressed disease but also promoted plant growth, increasing biomass and pseudostem diameter under pathogen pressure.
These findings support the use of native Trichoderma isolates as a sustainable strategy for Fusarium wilt management and as a preventive tool within integrated biosecurity programs.
Acknowledgments
The authors gratefully acknowledge the financial support provided by the DAPME project, a national initiative aimed at strengthening agricultural development and plant health programs, and by FONTAGRO, a regional cooperation mechanism that promotes innovation and research for agricultural development in Latin America and the Caribbean. We also sincerely thank Dr. Danilo Vera and MSc. Iván Garzón for their valuable technical support and scientific guidance throughout the development of this study. Special appreciation is extended to the Biotechnology Department of the Tropical Experimental Station Pichilingue (INIAP) for providing plant material and logistical support essential to the execution of this research.
References
Anis Mufida, D. R., Putra, I. P., Nawangsih, A. A., Ratna Ayu Krishanti, N. P., & Wahyudi, A. T. (2024). Glucanase enzyme activity from rhizospheric Streptomyces spp. inhibits growth and damages the cell wall of Fusarium oxysporum. Rhizosphere, 32, 100991. https://doi.org/10.1016/j.rhisph.2024.100991
Chávez-Avilés, M. N., García-Álvarez, M., Ávila-Oviedo, J. L., Hernández-Hernández, I., Bautista-Ortega, P. I., & Macías-Rodríguez, L. I. (2024). Volatile organic compounds produced by Trichoderma asperellum with antifungal properties against Colletotrichum acutatum. Microorganisms, 12(10), 2007. https://doi.org/10.3390/microorganisms12102007
Chen, J., Zhang, H., Zhang, X., Tang, M., et al. (2021). Antagonistic activity of Trichoderma spp. against Fusarium oxysporum triggers host defense responses. Microorganisms, 9(4), 717.
Dita, M., Echegoyen, P., & Pérez, L. (2017). Plan de contingencia ante un brote de Raza 4 tropical de Fusarium oxysporum f. sp. cubense en un país de la región del OIRSA. Organismo Internacional Regional de Sanidad Agropecuaria (OIRSA).
Esquivel-Naranjo, E. U., Mancilla-Díaz, H., Márquez-Mazlín, R., Alizadeh, H., Kandula, D., Hampton, J., & Mendoza-Mendoza, A. (2025). Light regulates secreted metabolite production and antagonistic activity in Trichoderma. Journal of Fungi, 11(1), 9. https://doi.org/10.3390/jof11010009
Fotoohiyan, Z., Rezaee, S., Bonjar, G. H. S., Mohammadi, A. H., & Moradi, M. (2017). Biocontrol potential of Trichoderma harzianum in controlling wilt disease of pistachio caused by Verticillium dahliae. Journal of Plant Protection Research, 57(2), 185–193. https://doi.org/10.1515/jppr-2017-0025
Harman, G. E., Doni, F., Khadka, R. B., & Uphoff, N. (2021). Endophytic strains of Trichoderma increase plants’ photosynthetic capability. Journal of Applied Microbiology, 130(2), 529–546. https://doi.org/10.1111/jam.14368
Izquierdo-García, L. F., Carmona-Gutiérrez, S. L., Moreno-Velandia, C. A., Villarreal-Navarrete, A. del P., Burbano-David, D. M., Quiroga-Mateus, R. Y., Gómez-Marroquín, M. R., Rodríguez-Yzquierdo, G. A., & Betancourt-Vásquez, M. (2024). Microbial-based biofungicides mitigate the damage caused by Fusarium oxysporum f. sp. cubense Race 1 and improve the physiological performance in banana. Journal of Fungi, 10(6), 419. https://doi.org/10.3390/jof10060419
Jamil, M. (2021). Antifungal and plant growth-promoting activity of Trichoderma spp. against Fusarium oxysporum f. sp. lycopersici colonizing tomato. Journal of Plant Protection Research, 61(3), 292–301. https://doi.org/10.24425/jppr.2021.138373
Kema, G. H. J., Drenth, A., Dita, M., Jansen, K., Vellema, S., & Stoorvogel, J. J. (2021).Fusarium wilt of banana, a recurring threat to global banana production. Frontiers in Plant Science, 11, 628888. https://doi.org/10.3389/fpls.2020.628888
Khuong, N. Q., Trang, C. T. T., Xuan, D. T., Quang, L. T., Huu, T. N., Xuan, L. N. T., Sakagami, J.-I., & Thuc, L. V. (2023). Evaluation of the antagonistic potential of Trichoderma spp. against Fusarium oxysporum F.28.1A. Journal of Plant Protection Research, 63(1), 16–26. https://doi.org/10.24425/jppr.2023.145993
Kumari, R., Devi, S., et al. (2024). Screening of biocontrol efficacy of Trichoderma isolates and their antifungal metabolites. Journal of Fungi, 10(6), 408.
Luo, M., Chen, Y., Huang, Q., Huang, Z., Song, H., & Dong, Z. (2023). Trichoderma koningiopsis Tk905 induces resistance and promotes growth in banana affected by Fusarium wilt. Frontiers in Microbiology, 14, 1301062.
Natsiopoulos, D., Topalidou, E., Mantzoukas, S., & Eliopoulos, P. A. (2024). Endophytic Trichoderma: potential and prospects for plant health management. Pathogens, 13(7), 548. https://doi.org/10.3390/pathogens13070548
Nguyen Quoc Khuong, N., Pham Van Kim, P., Le Thanh, T., & Tran Thi, T. (2023). Evaluation of the antagonistic potential of Trichoderma spp. against Fusarium oxysporum. Journal of Plant Protection Research, 63(2), 203–213. https://doi.org/10.24425/jppr.2023.145874
Ploetz, R. C. (2015). Management of Fusarium wilt of banana: a review with special reference to Tropical Race 4. Crop Protection, 73, 7–15. https://doi.org/10.1016/j.cropro.2015.01.007
Rodríguez-García, D., Patiño-Ruiz, J. D., et al. (2020). In vitro effectiveness of Trichoderma spp. against Fusarium oxysporum. Agronomía Mesoamericana, 31(2), 109–123.
Sarma, M., Zorrilla-Fontanesi, Y., Uma, S., Vanderschuren, H., Swennen, R., & De Coninck, B. (2025). Suppression of Fusarium wilt and growth promotion in banana by Trichoderma asperellum is cultivar dependent. Biological Control, 194, 105318.
Sánchez-Alarcón, J., Milić, M., Kašuba, V., Tenorio-Arvide, M. G., Montiel-González, J. M. R., Bonassi, S., & Valencia-Quintana, R. (2021). A systematic review of studies on genotoxicity and related biomarkers in populations exposed to pesticides in Mexico. Toxics, 9(11), 272. https://doi.org/10.3390/toxics9110272
Silva, A. (2018). Acción in vitro de Trichoderma spp. y Bacillus spp. como controladores biológicos conjuntos contra Fusarium oxysporum en uvilla (Physalis peruviana), ecotipo colombiano, en la sierra norte y centro del Ecuador. Tesis de grado, Pontificia Universidad Católica del Ecuador, Ecuador.
Terrero Yépez, P. I., Factos, N., Rodulfo, P., Solis, K., Molina, C., & Mayorga, K. R. (2025). Trichoderma spp. y su influencia en la resiliencia de plantas de plátano ante Ralstonia solanacearum (Smith) filotipo II. Siembra, 12(1), e7943. https://doi.org/10.29166/siembra.v12i1.7943
Torres-Rodríguez, J. A., Reyes-Pérez, J. J., Adame, L. H., Llerena-Fuentes, B. L., & Hernández-Montiel, L. G. (2024). Marine actinomycetes for biocontrol of Fusarium solani in tomato plants: in vitro and in vivo studies. Notulae Botanicae Horti Agrobotanici Cluj-Napoca, 52(2), 13562. https://doi.org/10.15835/nbha52213562
Xue, C., Penton, C. R., Shen, Z., Zhang, R., Huang, Q., Li, R., & Shen, Q. (2022). Manipulating the banana rhizosphere microbiome for biological control of Panama disease. Communications Biology, 5, 259. https://doi.org/10.1038/s42003-022-03238-2
Yao, X., Chen, Z., Xu, Y., et al. (2023). Trichoderma spp. in plant disease control and growth promotion. Frontiers in Microbiology, 14, 1160551.
Zheng, R., Wang, D., Li, X., Yang, M., Kong, Q., & Ren, X. (2024). Screening of core microorganisms in healthy and diseased peaches and effect evaluation of biocontrol bacteria (Burkholderia sp.). Food Microbiology, 120, 104465. https://doi.org/10.1016/j.fm.2024.104465
Zou, Y., Liu, Z., Chen, Y., Wang, Y., & Feng, S. (2024). Crop rotation and diversification in China: enhancing sustainable agriculture and resilience. Agriculture, 14(9), 1465. https://doi.org/10.3390/agriculture14091465